In the classically aloof terminology of his time, 17th-century German physician Samuel Hafenreffer described pruritis, or itch, as an “unpleasant sensation that elicits the desire or reflex to scratch.” This definition of pruritis is perhaps an understatement. In reality, chronic itch diseases, like atopic dermatitis, can be debilitating. Often, chronic itch is so severe that it can drive profound psychiatric sequelae – studies show that one-third of pruritus patients have comorbid psychiatric disorders as well as significantly elevated levels of suicidal ideation and anxiety. Indeed, itch is so pervasive that you may be feeling slightly itchy right now while reading this.
A central issue of chronic itch is that it remains largely intractable. Treatments are still based on “home remedies” like ointments, baths, and medications which, like antihistamines and steroids, are largely ineffective. A reason for these poor treatment options is the lack of effective drug targets.
A new study from Pang-Yen Tseng and Mark Hoon at the National Institutes of Health, Bethesda, US, has uncovered a new key player in inflammatory itch disease, and is raising hopes for a potential new antipruritic drug target. OSM is highly upregulated in the skin of patients with chronic itch diseases, and blocking OSM receptors on sensory neurons strongly attenuated itch in a mouse model. While OSM was not found to be a direct pruritogen, its release from immune cells potentiated neuronal responses to itch signaling.
The study was published in Science Translational Medicine on 10 November 2021, and is part of a series of papers from the Hoon lab that aim to question if mouse models of chronic itch have translational relevance in human disease. A review and original research paper are published, and more are currently under review.
OSM is enriched in itch-sensitive neurons
Historically, itch was thought about purely in relation to sensory neuronal activation, but recent evidence shows that itch signaling in the skin is an incredibly complex process involving multiple cell types.
“At some point, almost all cell types in the skin get involved in itch – neurons, immune cells, even skin cells like keratinocytes release pruritogenic compounds when they are damaged. You can think of itch like a bicycle wheel with cyclical interactions between neurons and other cells amplifying that signaling. Ideally, we could target molecular pathways in any cell type to stop the 'wheel' and reduce itch,” commented Mark Hoon.
The authors began by focusing on one cell type – neurons – and branching out to other “spokes on the wheel” from there. Hoon’s lab had previously shown that a small population of neurons expressing neuropeptide Nppb (natriuretic polypeptide b) are primary pruriceptive neurons, and can directly drive chronic itch behaviors in mice. The identity of pruritogenic agents in the skin that activate Nppb-expressing neurons remains unclear; however, cytokines are known to be important for inflammatory itch signaling. Studies have shown that cytokines, like interleukin-31 (IL-31), can directly depolarize sensory neurons and drive pruriception.
The authors decided to profile the expression of cytokine receptors in cutaneous sensory neurons. Initially, RNA-sequencing was performed in mice, specifically in Nppb-expressing neurons.As expected, the analysis revealed enrichment of IL-31 receptor and oncostatin M receptor (OSMR), as well as Janus kinase 1 (JAK1), molecules known to be crucial for cytokine signaling. Previously, OSMR was thought of as a co-receptor for IL-31 signal transduction. However, RNA-sequencing revealed that sensory neurons also highly express gp130, the co-receptor for OSM signal transduction.
OSM is highly present in inflammatory itch diseases
These high expression levels of OSM receptor complex in Nppb-expressing neurons suggested an important role for OSM in itch signaling; however, these initial experiments were performed in mice. The authors were keen to show translationally relevant data in the study.
First author Tseng told PRF that they “… wanted to focus on human data here. There are key molecular differences between mouse and human dorsal root ganglia [DRG], and if the itch-related molecules are not present in humans, then they’re not really important. Our main goal was to find molecular targets to alleviate chronic itch.”
“This paper also raises the question, Are the right molecules being looked at in itch and pain? This is especially true when translational data comes into play, as there are significant differences between mouse and human DRGs. Another big challenge of the pain field is what are the relevant molecules for us, not for mice,” said Hoon.
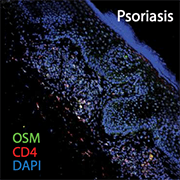
To explore this issue, the authors performed ISH-studies (in situ hybridization) using human DRG neurons. They found similarly high expression of OSMR selectively in human Nppb-expressing neurons. These results were promising, but the authors were keen to relate OSM expression to chronic itch diseases. Here, the authors examined RNA-sequencing datasets from the skin biopsies of three patient groups presenting with different pruritic skin diseases: psoriasis, atopic dermatitis, and cutaneous T cell lymphoma. The datasets revealed that OSM is highly upregulated in the skin from all patient groups, suggesting a common itch-related factor in these heterogeneous diseases.
“Just like there are multiple types of pain, there are multiple types of itch. What we aren’t saying is OSM is involved with all itches; rather, it seems to be very important in these three most prominent itch diseases,” offered Hoon.
OSM potentiates itch signaling but is not a pruritogen
Since OSM was upregulated in inflammatory itch conditions, the authors wanted to investigate whether OSM activated neurons as a pruritogen. Here, they turned to in vitro calcium imaging and whole-cell electrophysiology experiments to assess whether the application of OSM directly to mouse Nppb-expressing neurons would drive their depolarization and activation. These Nppb-expressing neurons did not respond to OSM, and there was no suggestion of calcium transience or depolarizations.
“These results were not entirely unexpected,” said Tseng. “Cytokines don’t directly gate ion channels and depolarize neurons; rather, they are coupled to signaling pathways involving kinases and transcription factors to induce protein phosphorylation or turn on gene expression. We thought that OSM might prime neurons in this way instead.”
With this hypothesis, the authors applied histamine to DRG neurons to observe if OSM would alter neuronal activation. OSM application alone did nothing; however, when they applied histamine shortly after OSM pre-treatment, it strongly potentiated histamine-induced neuronal activation. These findings are one of the most important messages of this paper, and show that while OSM is not the direct pruritogen, it acts to potentiate itch and accelerate the “bicycle wheel” previously described by Hoon.
Immune-neuronal signaling: OSM is released from T cells
While OSM is released in the skin to potentiate neuronal itch signaling via OSMR, the next logical step was to understand which cells are secreting OSM. Finding these cell types, however, would not be easy, as almost any cell type in the skin could be the source of OSM. The authors detected an early clue in single-cell RNA-sequencing datasets gathered from patients with cutaneous T cell lymphoma or with psoriasis. Here, the researchers saw that OSM was mainly expressed in CD45+ leukocytes, which they narrowed down to T cells and monocytes. Immunohistochemistry performed on the skin biopsies taken from patients with psoriasis also showed a strong presence of OSM in those immune cells that infiltrated inflammatory skin.
“Overall, these experiments show that OSM is released from T cells and monocytes. Both immune cells are a bit under the radar in itch signaling, but I think that these two cell types could be more important than other cell types, like mast cells, during chronic skin inflammation; this will be the focus of my K22-funded future work,” said Tseng.
Is OSM a new target for inflammatory itch treatment?
So far, these results pointed to OSM as a crucial itch-sensitizing molecule. The authors hypothesized that OSM is released from immune cells by an unknown pruritogen which sensitizes itch-selective neurons via the OSM receptor complex. This points to OSM as a key driver of chronic itch, and its inhibition could be a highly effective treatment.
In a final set of experiments, the authors tested whether inhibiting OSM signaling reduces inflammatory itch in mice. Here, they used a mouse model of inflammatory skin pruritis – psoriasiform dermatitis, induced in mice after repeated imiquimod treatment – resulting in intense scratching behavior at the site of treatment. Blocking OSM signaling was performed using SC-144, a drug that inhibits the OSMR/gp130 receptor complex. As expected, administering SC-144 greatly reduced scratching behavior and mitigated skin inflammation, providing promising early evidence that blocking OSM signaling might be an effective treatment for chronic skin inflammatory diseases. Indeed, Amanda Klein, a group leader at the University of Minnesota, US, who was not part of the study, highlighted the potential link between OSM signaling and a new set of drugs that are proving efficacious in treating inflammatory itch in early drug development trials.
“Janus kinase [JAK] inhibitors are a somewhat newer class of drugs approved for rheumatoid arthritis. Now they are in development for chronic itch, and are showing promising results in early phase clinical trials. The RNA-sequencing data in this paper show a possible link between inflammatory skin diseases and OSM. JAKs appear to be important for OSM signaling, so maybe the OSM is part of the story as to why JAK inhibition might be effective for treating chronic itch. This opens up a whole new direction looking at various kinase inhibitors for treating itch,” said Klein.
Let’s hope that targeting OSM signaling could be a new approach for treating inflammatory itch conditions. It would certainly be more effective than current remedies such as wearing oven mitts to stop scratching.
Fred Schwaller, PhD, is a freelance science writer based in Germany.
Image: Representative image of a parafilm-embedded human skin section immunostained against OSM (green) and CD4 (red) in a psoriatic skin sample. Tseng and Hoon. Science Translational Medicine. 2021 Nov; 13.