Millions of people continue to misuse opioids each year, pointing to the need for an improved understanding of the determinants of these painkillers' detrimental effects. A recent study now proposes a new contributor to opioid addiction and in doing so points one way forward to address the opioid crisis.
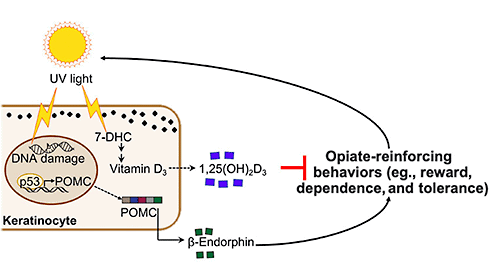
The research from David Fisher, Harvard Medical School, Boston, US, and colleagues reveals an association between opioid use/opioid use disorder (OUD) and low vitamin D levels in people. Further, the investigators show in mouse models that deficient vitamin D signaling amplifies responses to opioids, including the reward, analgesia, tolerance, and dependence associated with use of these drugs. Further, restoring vitamin D signaling normalized responses to morphine in the animals. These findings point to a focus on vitamin D as one possible avenue to ameliorate the opioid crisis.
“We are living in an opioid overdose crisis with limited solutions, so this is an important topic. The relapse rate is really high for people with an opioid addiction, and there is a huge need to develop novel therapies. This study is fascinating, as it points out links between vitamin D, OUD, and morphine effectiveness,” said Anna Taylor, University of Alberta, Edmonton, Canada, who was not involved in the study.
The findings appeared June 11, 2021, in Science Advances.
Uncovering an association
Initially, the researchers were trying to understand the biological causes of harmful sun-seeking behaviors, which can lead to premature skin aging and increased risk of skin cancer. That work paved the way for discovering the role of vitamin D in opioid responses and addictive behaviors.
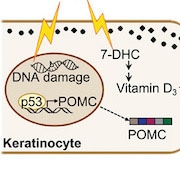
A previous study from the group found that ultraviolet (UV) radiation can spur skin synthesis of pro-opiomelanocortin (POMC), a precursor for the skin pigment melanin, and for β-endorphin, an endogenous opioid (Fell et al., 2014). This and other studies led to the researchers' current study hypothesis.
“We thought that people may be seeking sunlight to avoid feeling bad, as a result of not getting enough vitamin D and endogenous opioids, so we posited that UV-seeking behaviors serve to maximize vitamin D synthesis, and that vitamin D deficiency may intervene with reward signaling to potentiate opioid use,” Fisher said.
The group first asked whether there was a correlation between opioid use and serum vitamin D levels in people. Using data from the National Health and Nutrition Examination Survey (NHANES, 2003-2004), the group found that people with deficient (<12 ng/mL) or insufficient (<20 ng/mL) vitamin D levels were more likely to use opioid painkillers (odds ratios of 1.62 and 1.27, respectively). The odds ratios were even higher when controlling for confounding factors like age and the presence of chronic pain (odds ratios of 1.9 and 1.52).
Since higher opioid intake can increase the risk of OUD, the investigators also looked into any possible association between vitamin D deficiency and OUD. In a cohort of primary care patients at Massachusetts General Hospital from the years 2014 to 2016, patients with OUD (N = 2,772) were more likely to have vitamin D deficiency compared to age-, sex-, race-, and primary care provider-matched controls (control N = 8,265, 28% vs. 17%).
Vitamin D deficiency increases sensitivity to morphine in mice
Based on the association between vitamin D levels and opioid use in humans, the group turned to mouse models to understand how vitamin D deficiency might affect responses to opioids. To model vitamin D deficiency, mice were fed a diet without vitamin D for at least eight weeks. Some mice returned to a normal diet for eight weeks to restore their vitamin D levels. To model deficient vitamin D signaling, the researchers also included vitamin D receptor knockout mice in the study.
In a conditioned place preference paradigm, mice were allowed free movement between two chambers, one of which was paired with morphine and the other with vehicle. While control mice most preferred the morphine-paired chamber only at the highest dose of the drug, the vitamin D-deficient and vitamin D receptor-deficient animals spent more time in the morphine-paired chamber at all doses tested in these experiments. Importantly, restoration of vitamin D levels through oral supplementation normalized this increased preference for morphine.
To learn more about the effects of vitamin D deficiency on other behaviors seen with opioid use, the researchers next turned to the hot plate test to interrogate thermal nociception. Basal thermal nociceptive thresholds were higher in vitamin D- and vitamin D receptor-deficient mice, which depended on the presence of mu-opioid receptors (MORs), according to experiments with MOR knockout mice. The higher thresholds were rescued by oral vitamin D. Additional experiments revealed that the effect of decreased vitamin D signaling on nociceptive thresholds depended on central opioid signaling.
The investigators also saw enhanced morphine analgesia in vitamin D- and vitamin D receptor-deficient mice, compared to wild-type animals, along with quicker tolerance to morphine upon repeated exposure to the drug. The authors then used naloxone to produce withdrawal to morphine, in order to assess the contribution of vitamin D signaling to drug dependence. Morphine dependence was greater in vitamin D- and vitamin D receptor-deficient mice. Similar to the results from the place preference experiments, these other morphine responses were normalized by restoring vitamin D levels through the diet. Together, these behavioral observations supported the idea that vitamin D deficiency increases opioid responses.
A role for the brain's reward pathway
Next, the researchers sought to identify the mechanism behind the enhanced morphine reward in vitamin D receptor-deficient mice, using RNA sequencing. Compared to wild-type animals, the deficient mice exhibited an increase in cFos, a transcription factor that is an indirect marker of neuronal activation, in the nucleus accumbens (NAc), a brain region known for its role in morphine reward, and in the ventral tegmental area (VTA), another key component of the brain's reward pathway, particularly via dopamine signaling.
Further, “using RNA sequencing, we also saw downregulation of genes involved in dopamine signaling in the VTA, such as monoamine oxidase A and catechol-O-methyltransferase, in vitamin D receptor-deficient mice, implicating a role of vitamin D in regulating dopamine metabolism,” according to Lajos Kemény, co-first author of the study along with Kathleen Robinson. Dopaminergic signaling between the VTA and NAc is a key pathway mediating opioid-related reward (Arias-Carrión et al., 2010).
The study also found an inverse correlation between vitamin D receptor messenger RNA (mRNA) and MOR mRNA in the human VTA. This was in line with the observations in mouse models that vitamin D deficiency increases opioid signaling.
A final set of experiments would show that vitamin D deficiency also increased endogenous analgesia and reward in response to UV light.
A new way forward to address the opioid crisis?
Together, the new data suggest that vitamin D should be studied as a potentially safe and effective therapy to normalize opioid responses, perhaps reducing the risk of addiction.
“We encourage people to use vitamin D supplements instead of seeking UV light for maintaining their vitamin D levels,” said Fisher. To confirm the utility of vitamin D in OUD, he added that “one clinical trial design could be to test and correct vitamin D levels in post-surgical pain patients, who rely on opioids for pain management, and ask whether patients develop the need to remain on opioids.”
In addition, “Vitamin D levels can be a biomarker that, once validated, presents a clinically meaningful way to screen patients for risk of developing OUD,” according to Taylor.
Charlie Kwok, PhD, is a postdoctoral fellow at Hotchkiss Brain Institute, University of Calgary, Canada.