Nociceptive signals entering the brain via the spinothalamic pathway allow us to detect the location and intensity of a painful sensation. But, at least as importantly, nociceptive inputs also reach other brain regions that give pain its emotional texture. Key to that circuitry is the parabrachial nucleus (PBN), a tiny cluster of cells in the brainstem associated with homeostatic regulation – of things like temperature and food intake – and the response to aversive stimuli and perceptions of many kinds. Now, two new papers advance the understanding of the role of the PBN in pain.
First, Asaf Keller and colleagues at the University of Maryland, Baltimore, US, show in both rats and mice that the PBN receives inhibitory inputs from the central nucleus of the amygdala (CeA), and that those inputs are diminished in chronic pain conditions, leading to PBN hyperactivity and increased pain perception. The inhibitory CeA neurons were distinguished by their expression of neurochemicals including dynorphin, somatostatin, and corticotropin-releasing hormone. The authors suggest that disinhibition of the amygdalo-parabrachial pathway may be crucial to establishing chronic pain.
Mary Heinricher, a pain researcher at Oregon Health & Science University, Portland, US, who was not involved in the study, called it “a real tour de force and a notable addition to the field. It has implications for how we think about nociceptive processing,” she said.
The paper was published April 22 in the Journal of Neuroscience.
A second paper, from Sarah Ross and colleagues at the University of Pittsburgh, US, investigated the efferent outflow from the PBN and its role in pain. That study, published June 17 in Neuron, identified distinct populations of PBN neurons projecting to separate target regions in the brain to mediate pain escape behaviors and aversive learning. The investigators also characterized a population of excitatory neurons that originated and terminated within the PBN itself, and conveyed noxious input from the dorsal division of the PBN to the lateral PBN.
Fusao Kato, a neuroscientist at Jikei University School of Medicine, Tokyo, Japan, who was not involved in either study, said he was “really excited reading both these papers.” Of the work from Ross and colleagues in particular, he said that “the most surprising and striking evidence that this paper found was identification of the dorsal part of the PBN as the first receiver of [spinal nociceptive input], which then transmits it to the CeA; this is quite new. So the nociceptive information that the CeA receives has already been processed at the level of the PBN.”
In recent years, distinct subpopulations of cells in the PBN have been shown to regulate behaviors associated with potentially threatening or noxious stimuli (see PRF related news). Work from Richard Palmiter at the University of Washington, Seattle, and others, Kato said, was “the beginning of a sort of boom of studies in the PBN. These papers are in line with this new wave of PBN studies.”
Loss of inhibition
In the first study, first author Charles Raver, a research associate in the Keller lab, used chronic constriction injury of the infraorbital nerve (CCI-ION) to model chronic pain in mice. Three weeks after surgery, the animals displayed a dramatically reduced mechanical withdrawal threshold, and a much higher grimace score, indicating the animals had evoked as well as ongoing pain.
The group had previously shown in rats that PBN activity was amplified following CCI-ION (Uddin et al., 2018). So in the current study, the researchers made electrophysiological recordings from individual PBN neurons and found that activity evoked by a face pinch was eightfold higher from CCI-ION neurons than those from sham-operated control mice. Curiously, about three-quarters of neurons from CCI-ION mice displayed after-discharges – an excitatory response that outlasts the stimulus – compared to only one in five neurons from sham-operated mice, and the after-discharges lasted significantly longer. Spontaneous firing rates were higher in PBN neurons from CCI-ION mice, too.
“The after-discharge is seen as an electrophysiological hallmark of chronic pain,” Raver said. “Cells are no longer responding in lockstep to stimulations in the receptive field but continue to fire long after the stimulus ends, suggesting a loss of inhibition – a feature of physiological changes in other regions of the brain in chronic pain.”
To further investigate that loss of inhibition, the group recorded from PBN neurons in acute slices and saw that miniature inhibitory postsynaptic currents (mIPSCs) occurred about half as frequently in neurons from CCI-ION mice compared to controls, but that their amplitudes were comparable.
GABAergic neurons, which provide inhibitory input, are relatively rare in the PBN of both mice and rats, so Raver looked to other sources of inhibition using anterograde and retrograde tracing. Injection of cholera toxin subunit B (CTB), a retrograde tracer, into the PBN in mice revealed robust inputs from the ipsilateral CeA, a region highly enriched in GABAergic neurons. Anterograde tracing from the CeA using a viral construct confirmed the pathway’s existence in mice.
The authors carried out similar experiments to demonstrate that the pathway was also present in rats. In addition to CTB, they also used fluorogold, a different retrograde tracer, to confirm the findings in both sexes of two different strains of rat. Viral anterograde tracing also confirmed the results.
“There were hints in the literature suggesting that the amygdala might provide inputs to the PBN, but not much else was known,” Keller said of the inputs from the CeA.
In both mice and rats, the investigators also saw some retrograde-labeled neurons in the zona incerta, the paraventricular and lateral hypothalamus, and the insular cortex.
Recent work has shown heterogeneity among CeA neurons (see PRF related news). To better understand the makeup of the CeA inputs to the PBN, Raver and colleagues used in situ hybridization to specific RNA target sequences, which revealed that the cells expressed dynorphin, somatostatin, corticotropin-releasing hormone (CRH), or some combination of the three neurochemicals – characteristics that distinguished the neurons among the CeA’s heterogeneous population.
A new pain pathway?
To investigate the functional consequences of CCI-ION on the CeA-PBN pathway, the authors next turned to optogenetics, expressing the excitatory light-sensitive protein channelrhodopsin-2 (ChR2) exclusively in GABAergic neurons of the CeA. Optical stimulation produced robust IPSCs in lateral PBN (lPBN) neurons in both uninjured and CCI-ION animals, but the amplitude of evoked IPSCs was on average five times larger in neurons from control animals compared to CCI-ION mice.
The group next measured the paired-pulse ratio (PPR), in which a pair of stimuli are delivered and the amplitude of the second evoked response is divided by the amplitude of the first. A low PPR is a reflection of synaptic strength, the authors explained, whereas a higher PPR is characteristic of a weaker synapse. The PPR was nearly twice as large in slices from CCI-ION mice compared to controls. Together, the reduced mIPSCs, smaller evoked IPSC amplitude, and higher PPR in CCI-ION neurons pointed to presynaptic changes in the CeA neurons terminating in the lPBN. The authors say that suggests a causal role for the pathway in chronic pain development.
To determine whether disinhibition affected pain behaviors, the researchers injected the lPBN of CCI-ION rats with the GABAA receptor agonist muscimol, which normalized mechanical withdrawal thresholds and facial grimace scores toward baseline. Saline injection had no such effect. In a corollary experiment, injection of the GABAA receptor antagonist gabazine in the lPBN of naïve rats led to freezing or escape behaviors, indicating it was aversive. Mechanical withdrawal thresholds also decreased in rats injected with gabazine bilaterally.
In addition to the pharmacological disinhibition experiments, the researchers optogenetically activated CeA axon terminals in the lPBN in awake animals, which led to greatly increased mechanical withdrawal thresholds and reduced responses to hindpaw brushing, which evokes dynamic mechanical allodynia. Following formalin injection, optical stimulation caused withdrawal thresholds to roughly double, and responses to brushing and facial grimace scores, both diminished significantly. The results suggest that activation of the CeA-PBN pathway reduces both ongoing and evoked pain.
Heinricher said that the experiments “reinforce the role of the PBN as a relay station that triggers plastic processes at multiple levels of the nervous system. One take-home is that the plasticity seemed to come in at the CeA-to-PBN synapse, not in the PBN itself.” It remains unclear, she said, where the plasticity is happening. “Is it in the amygdala? Or is something about that synapse special?”
Outputs From the PBN
In the second paper, first author Michael Chiang, a graduate student in the Ross lab, set out to explore the outputs from and spinal inputs to the lPBN.
The researchers began by expressing ChR2 in GABAergic neurons found in the lPBN of mice. Photostimulation of these inhibitory neurons attenuated capsaicin-induced mechanical hypersensitivity. Pain behaviors were similarly inhibited in mice with chemogenetic inhibition of lPBN excitatory neurons following acute capsaicin or prolonged complete Freund’s adjuvant (CFA). The animals also displayed a muted jumping response away from a heat source.
Further, while control mice displayed conditioned pain modulation in response to a noxious stimulus, as indicated by a blunted tail flick response, this was absent in mice subjected to chemogenetic inhibition. And the conditioned place aversion to a noxious stimulus seen in control mice was reduced in chemogenetically treated mice, suggesting that the lPBN mediates pain-related escape behaviors, the learning that ensues, and conditioned pain modulation.
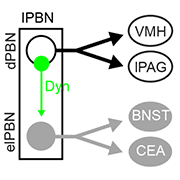
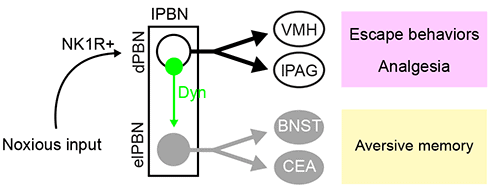
Chiang and colleagues next wanted to tease apart the cell populations and their downstream targets, regulating these different behaviors by using a fluorescent protein to visualize axon projections and presynaptic terminals from the lPBN. In line with previous observations, the efferents terminated in the bed nucleus of the stria terminalis (BNST), CeA, ventral medial hypothalamus (VMH), and lateral periaqueductal gray (lPAG). Most lPBN outputs were excitatory, though some projections were GABAergic.
Are lPBN efferents heterogeneous? Or do they make up a single output population to divergent targets? The researchers tackled this question by using retrograde tracing from the target regions and found that each target region was innervated by neurons in spatially distinct subregions of the lPBN. Double-labeling experiments revealed two main populations: Cells from the external lateral division of the PBN (elPBN) projected to the BNST and CeA, whereas neurons in the dorsal division (dPBN) projected to the VMH and lPAG.
Pain behaviors
To determine whether these two populations mediated different pain behaviors, the team next stimulated the lPBN using optogenetics, implanting optical fibers above the efferent target regions.
Perhaps not surprisingly, given the PBN’s role as an aversion center, photostimulation of any target region led the mice to spend more time in the non-stimulation area of a cage. Further investigation with a conditioned place aversion assay, however, showed that only stimulation of efferents to the CeA or BNST resulted in stable aversive learning.
Kato applauded the group’s use of multiple measures of pain. “Pain is an emotional as well as a sensory experience; mechanical allodynia is not enough to be called pain. Here they measured many other pain-related behaviors: place aversion, avoidance, and escape. That allowed them to dissect different pain-related behaviors in relation to distinct subnuclei of the PBN,” he said.
Inputs to the lPBN
Next, to elucidate the nociceptive inputs to the lPBN, the researchers fluorescently labeled neurokinin 1 receptor-expressing spinoparabrachial neurons, which transmit noxious signals from the spinal cord to the PBN. Most of the inputs to PBN were localized to the dPBN rather than the elPBN. Noxious stimulation of the hindpaw with capsaicin, however, led to increased expression of Fos, an indirect marker of neuronal activity, in both regions of the PBN.
Chiang next looked for neurons that could convey noxious signals within the PBN itself. Those experiments showed that only dynorphin-positive PBN neurons were poised to do so. Indeed, the dynorphin-positive neurons in the dPBN sent dense projections to the elPBN, far more than to other downstream targets. Optogenetic stimulation of spinoparabrachial terminals produced excitatory postsynaptic currents in the dynorphin PBN neurons, and intraplantar capsaicin injection led to strong Fos induction in those same neurons. Together, the results indicated that dPBN dynorphin neurons receive noxious stimuli via spinobrachial inputs, and then transmit those inputs to the elPBN. Nearly all the dynorphin PBN neurons were glutamatergic.
Finally, photostimulation of the dynorphin PBN neurons elicited aversive but not escape behaviors in mice, while selective ablation of these neurons resulted in a lack of conditioned place aversion while leaving conditioned pain modulation intact. The authors say that the dynorphin PBN neurons represent a crucial link between the dPBN and elPBN efferents targeting the CeA and BNST.
The dynorphin-positive neurons found in the two studies are different populations with distinct functions, Chiang said. “In the Keller study, the dynorphin neurons they see in the CeA are inhibitory, whereas we characterize the PBN dynorphin neurons as excitatory. Whether those two populations are interacting in any way is an interesting idea to explore.”
Keller was enthusiastic about many aspects of the Ross paper. The discovery that dynorphin neurons relay excitatory inputs within the PBN was particularly intriguing, he said. “This is one of the first intra-PBN microcircuits discovered. To understand input transformation and integration by this nuclear complex, we need to further elucidate its intrinsic circuitry,” he said.
Heinricher said there’s a big takeaway from the two studies. “This tells us that chronic pain is manufactured by the brain. It’s not a one-way process driven by something coming up from the periphery; the brain is not a passive recipient. To me that’s what’s exciting: The brain is actively constructing a chronic pain state in part by this recurring circuit.”
Kato agreed, saying that the field needs to think about pain networks as composed of distributed multiple layers at the spinal cord, brainstem, and higher levels, rather than as simple pathways.
“People used to think that the PBN was just a passive relay center, relaying nociceptive information up to other nuclei. This was simply because we did not know much about the PBN. The new concept here is that even within the PBN, this tiny nucleus, there could be synaptic modulation of information. So there could be some network integration or modification of nociceptive signals.”
The center of the universe?
Recent studies of the PBN by other groups (including recent work by Deng et al., 2020) have been revealing a role for this structure in pain, said Keller. “There have been hints that the PBN was important for pain over the years, but it was relegated to a curiosity. In the last several years, groups have started delving back into the PBN and showing it is indeed involved in acute and chronic pain, especially the affective, aversive aspects of pain. That’s coupled with the realization that most inputs from the spinal cord converge on the PBN rather than spinothalamic targets.”
The PBN does not react specifically to pain, though. “We know it’s involved in a number of different functions. The overarching theme is that the PBN is some kind of aversion center, not only for pain,” Keller said.
Ross agreed that the complex functions of the PBN are only now being elucidated. “The role of the PBN is to sound the alarm button” when an organism is in danger, but its role goes further. “Really it is better characterized as a key homeostatic center, weighing short-term versus long-term survival. If you’re warm, fed, and comfortable, for example,” organisms can address long-term directives like procreation. “When you’re unsafe, though, you need to put those things off and deal with the emergency situation,” she said.
Heinricher said the recent studies have led her to think that, when it comes to pain, the PBN “might be the center of the universe. It really is this incredible switching yard lurking in this one little tiny corner of the brainstem.”
Stephani Sutherland, PhD, is a neuroscientist and freelance journalist in Southern California. Follow her on Twitter @SutherlandPhD