Fibromyalgia syndrome (FMS) is one of the most common chronic pain conditions, and yet the underlying mechanisms driving it have remained elusive. Previous work has started to build a case for autoimmune mechanisms as a driver of pain, and now a new study further builds the case for autoimmunity as a pain culprit, this time in the context of FMS.
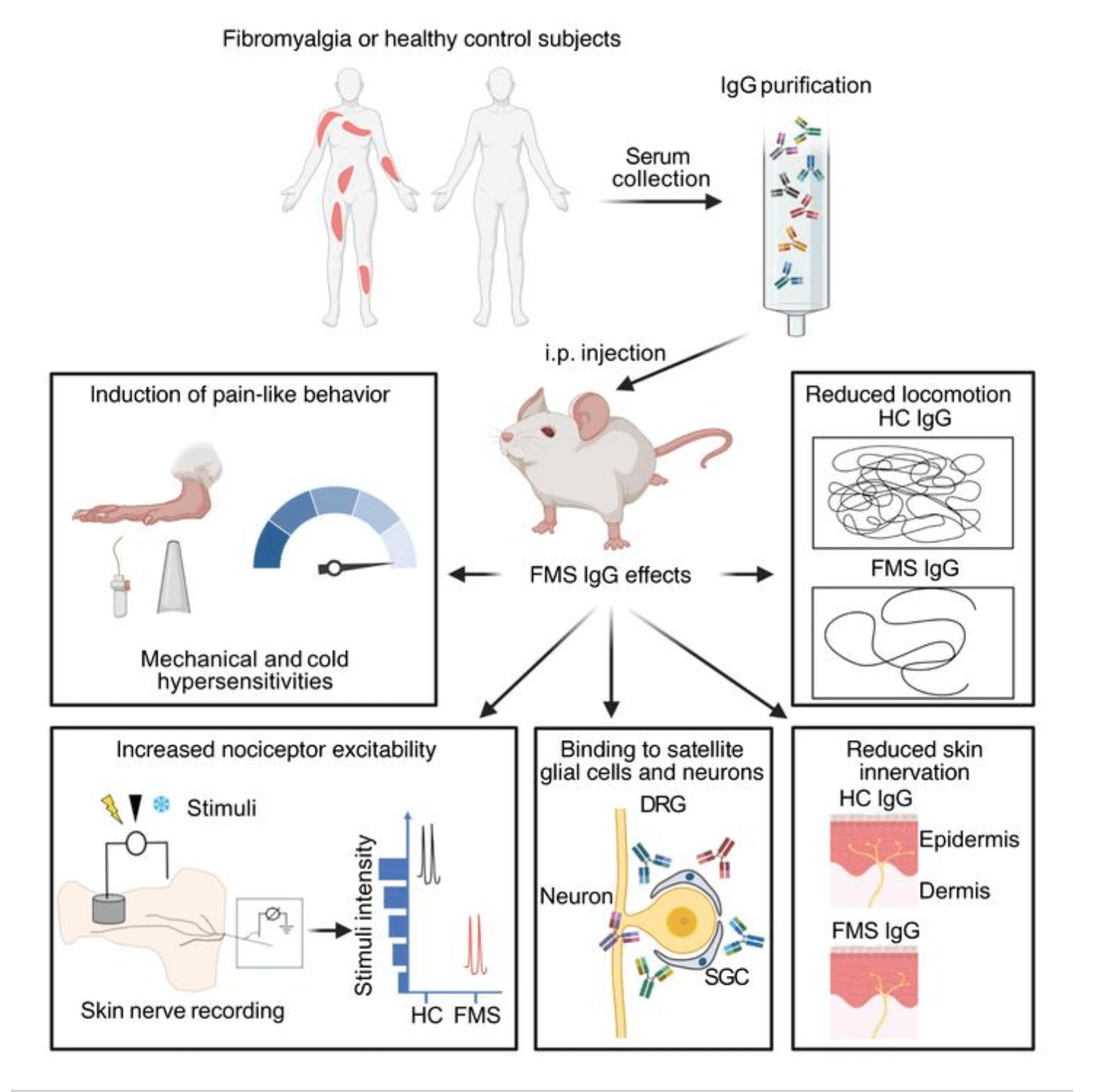
Research from co-senior authors David Andersson, King’s College London, UK, and Camilla Svensson, Karolinska Institute, Stockholm, Sweden; clinical lead author Andreas Goebel, Walton Centre NHS Foundation Trust and University of Liverpool, UK; and colleagues shows that antibodies from patients with FMS can cause fibromyalgia symptoms in mice. Specifically, injection of Immunoglobulin G (IgG) antibodies from FMS patients into mice increased mechanical and cold hypersensitivity in the animals by sensitizing nociceptive neurons. The findings also point to satellite glial cells as important cellular players in driving this hypersensitivity.
In addition, the FMS antibodies reduced locomotion and paw grip strength, and also caused a loss of intra-epidermal nerve fibers (IEF). These findings are relevant to FMS since fatigue, reduced handgrip strength, and IEF loss are each associated with the human condition.
The study “is a shining, impactful example of how and why clinical translational research can and should be done. This tour de force study at once provides insight into a mysterious disease affecting millions of people, and offers a solid and promising insight into just how to begin developing effective therapeutic strategies for a severe, chronic, uncurable illness,” wrote Kevin Tracey, Northwell Health, Manhasset, US, in an accompanying commentary.
However, ample caution in interpreting the current study results is warranted, according to Daniel Clauw, University of Michigan, Ann Arbor, US.
“My overall impression is very positive, but I would like to see if the results can be replicated, as it’s a different paradigm to what is known thus far with respect to the mechanism that is involved in FMS,” according to Clauw, a physician and researcher who studies chronic pain, including FMS pain, but who was not involved in the new work. “As a researcher I must be a bit skeptical until multiple other laboratories can confirm the same findings and the same mechanism of IgG driving the FMS phenotype seen in mice, especially since this is a cross-species study.”
The research and accompanying commentary were published July 1, 2021, in The Journal of Clinical Investigation.
From patients to mice
Andersson, Svensson, and Goebel all had a keen interest in autoantibodies, although in different pain conditions such as complex regional pain syndrome and rheumatoid arthritis (RA). The three of them met at the International Association for the Study of Pain (IASP) World Congress on Pain in 2016 in Yokohama, Japan, where they initiated a collaboration to look at the role that autoantibodies might play in FMS pathophysiology.
Because many patients with RA are also diagnosed with FMS, and research had just shown that autoantibodies were involved in the RA pain phenotype without a clear link to tissue inflammation, and as they had already demonstrated the importance of autoantibodies in a related chronic primary pain condition – chronic regional pain syndrome (CRPS) – the authors hypothesized that FMS might also have an autoimmune basis and thus an auto-antibody component (see PRF related interview with Goebel). FMS was also important to study, considering the state of knowledge about the condition.
“There is a pressing need to find underlying causes for the pain phenotype of FMS patients, as the origins still remain unknown,” said Andersson.
So the authors made use of IgG from FMS patients in both the UK and Sweden, and conducted the experiments in multiple laboratories. The investigators purified IgG from the serum of FMS patients or healthy controls and administered it to mice by intraperitoneal injection for four consecutive days.
The IgG from eight individual UK patients with FMS, but not from controls, rapidly (within 24-48 hours) produced mechanical hypersensitivity, as measured by the von Frey assay, in the mice. The FMS antibodies also increased the animals' cold sensitivity, a symptom often reported by FMS patients.
FMS IgG-treated mice also exhibited decreased front paw grip strength, as well as heightened pressure sensitivity as measured with the Randall-Selitto device, where the thigh is squeezed until the animal withdraws its paw or vocalizes.
The authors also examined the effect of a single dose of IgG and found that it was sufficient to elicit almost the same mechanical hypersensitivity as the four separate doses of IgG used in the earlier experiments.
Transfer of FMS symptoms from pooled samples
To strengthen the idea that IgG from the general FMS population can drive these behavioral phenotypes, the authors next used pooled IgG from 44 Swedish FMS patients, and pooled IgG from controls.
As expected, the researchers found that FMS patients had increased pressure pain and higher pain ratings compared to the healthy controls. Next, the pooled IgG was administered to mice. Similar to the data from individual patient IgG, pooled IgG from FMS patients evoked mechanical hypersensitivity in mice, but pooled control IgG did not. The pooled IgG-induced mechanical hypersensitivity followed the same time course as the mechanical hypersensitivity in response to individual patient IgG. The hypersensitivities always fully resolved two to three weeks after the last IgG injection.
“It was very surprising that the mechanical hypersensitivity phenotype, which can vary quite a bit, was so reproducible from individual-to-individual sample and also in the pooled samples in these mice,” said Goebel.
The group also saw that mice injected on two consecutive days with FMS IgG displayed a marked reduction in locomotor activity during the night, when mice are normally most active.
At the cellular level
Previous data indicated that C-fibers in FMS patients are hyperexcitable (Serra et al., 2014). To investigate whether this was also true of nociceptive fibers in the hindpaw skin of mice that received IgG, the authors performed skin-saphenous nerve recordings.
Similar to the earlier human studies, the authors found that C-mechanosensitive nociceptors, as well as A-delta mechanosensitive fibers, from FMS IgG-injected mice had reduced mechanical firing thresholds compared to controls.
The authors also found via Western blot experiments that FMS IgG was consistently observed in the dorsal root ganglion (DRG) but, interestingly, not in the spinal cord or brain. Immunohistochemistry with anti-IgG antibodies revealed robust staining in the lumbar DRG but not in the spinal cord of FMS IgG mice, compared to control IgG-injected mice or saline controls.
The group also found that the staining was primarily localized to satellite glial cells (SGCs) and some neurons. They further saw that FMS IgG promoted the expression of SGC activity markers such as glial fibrillary acidic protein (GFAP) and Gfap and s100b. These changes occurred only in the DRG, with no changes in astrocyte or microglia reactivity in the spinal cord.
SGCs wrap around neuronal cell bodies in the DRG, allowing for bidirectional communication between the two cell types. SGCs also release molecules that can stimulate nociceptors, such as ATP, glutamate, and cytokines. But there was no increase overall in circulating cytokines in the serum; nor was there systemic inflammation after FMS IgG injections into the serum of mice. Together, the results suggested that the FMS IgG effect is local, being restricted to the DRG.
Because small-fiber pathology, particular the dying back of nerve endings in the skin, is seen in FMS, the authors confirmed that FMS IgG injection decreased intra-epidermal nerve fiber density in the glabrous skin of mice 14 days after FMS IgG was initiated.
Finally, after the mouse experiments, the investigators set their sights on the human situation.
“Because, until this point, all of our research was focused on mice, we really wanted to make sure that our work would be translational, and thus we stained human DRG tissue samples with FMS IgG,” said co-first author Emerson Krock.
Similar to the mouse immunohistochemistry findings, FMS IgG bound to immunoreactive cells expressing GFAP or NF200 (a neuronal marker) in human DRG.
“For me, this was really the key piece of evidence to bring this whole story together, as it demonstrates translatability into humans,” stressed Krock.
A healthy dose of skepticism?
As for Clauw, he thinks that the mechanism identified in the current study leaves a lot about FMS unexplained.
“I am a bit skeptical, as any scientist should be, because it is difficult to understand how this auto-antibody mechanism could cause the rest of the pathophysiology we see in patients with FMS,” Clauw said.
“Patients with FMS do not just experience widespread pain, but they have a lot of other sensory sensitivities including sensitivity to light or odors, which are part of the nociplastic pain of FMS, as well as memory and sleep problems that in my opinion can simply not be explained by this peripheral mechanism where an IgG antibody binds to something in the DRG,” Clauw continued.
“These symptoms are not secondary to pain, and I have a hard time picturing how the effects seen here in the DRG affect the sensory processing in the spinal cord. Overall, I am looking for a mechanism that would allow me to explain all the FMS symptoms together, rather than just the pain and nociception symptoms,” said Clauw.
The authors agree with Clauw that the mechanisms underlying those other sensory sensitivities in FMS are unclear and may not have an IgG mechanistic basis in the DRG. But they pointed out that those sensitivities were not the focus of their study and are instead questions for future investigations to explore.
The response from FMS patients
The authors say they are touched by the responses they have received to their work from people with FMS.
“Since the paper came out, I have received between five to 10 emails every day from people who I never met, who describe their story and how they are struggling with FMS symptoms, and express how studies like ours give them hope. It is heartbreaking at times, and it certainly gives me and my team even more energy and motivation to continue with our research centered on fibromyalgia,” said Svensson.
Patients may also feel validated by the findings, according to Goebel.
“We were able to track part of the origin of a disease that is often called a ‘disease of the mind’ – perhaps in part because to this date it did not have a clear pathology. Finding a biological cause of pain is huge for the patient as well as the field; patients seem to feel more understood and validated. This could also apply to other disease states, such as tinnitus, where patients often do not feel understood either,” Goebel told PRF.
In terms of next steps, there are plenty of things for the researchers to pursue.
“We of course want to understand the cellular mechanism and the link between the autoantibodies and the satellite glial cells, and how this is driving the sensitization and increases in the excitability of nociceptors. We are very excited about the SGC findings but want to know if there are other cellular targets that these autoantibodies are binding to,” said Svensson.
Svensson continued, “My lab also finds the decreased nerve innervation in the skin very interesting. This effect occurred so rapidly, and we are asking whether these autoantibodies cause this localized effect through actions in the skin or if it is caused by changes in the DRG.”
“While we obviously see signs of muscle fatigue and weakness in the FMS IgG-treated mice in our study,” said Andersson, “another hallmark of FMS is cognitive fatigue. This is very hard to examine in mice, but we would like to determine if that could also be impacted by autoantibodies.”
As for Goebel, “I would also like to look at it from the complex regional pain syndrome perspective and investigate whether satellite glial cells might play a role in that, too, and use approaches similar to what we utilized here, to drive research in CRPS.”
Finally, the group hopes its work will lead to new treatments for FMS.
“It is of course implied that autoantibodies bind to something, and identifying the target is going to be the key that might lead to therapeutic options,” said Andersson. “If we can at least prevent the hypersensitivity of nociceptors, we could at least alleviate some of the pain that these patients are experiencing.”
“Additionally, even without knowing what the specific targets are, we may already be able to progress clinical therapies. Several existing therapies can reduce serum autoantibodies and are effective in established antibody-mediated disorders. We will now need to obtain the necessary funding so that we can start exploring their efficacy in FMS,” concluded Goebel.
Francie Moehring is a freelance writer based in Milwaukee, US.


