Researchers know that infectious agents can lead to pain either through direct or indirect activation of nociceptors. Nociceptors then release neuropeptides from their peripheral terminals, which in turn can influence the function of other cells, such as immune cells, present in the tissue. Through this mechanism, sensory neurons can regulate the immune response to bacterial and fungal pathogens.
Now, a new study led by Sophie Ugolini and colleagues at Aix Marseille University, France, demonstrates that sensory neurons also play a role in the immune response to viral infections in the skin. In particular, the work shows that nociceptors regulate both the innate and adaptive immune response to infection with herpes simplex virus 1 (HSV-1), an important human pathogen.
“This is one of the first studies showing the role of sensory neurons in regulating skin immune cells in response to viral infection,” said Isaac Chiu, a neuroimmunologist at Harvard Medical School, Boston, US. “The variety of neurobiological and viral techniques employed in the study provides strong evidence for the involvement of nociceptors in mediating the immune response to HSV-1 infection. Parts of this study may relate to previous findings in bacterial infections where neutrophils were shown to be suppressed by nociceptors, but a novel aspect is neural regulation of cytotoxic T cells,” according to Chiu, who was not involved in the research.
The work appeared 18 May 2021 in Nature Communications.
How it all started
Ugolini, an immunologist, became interested in exploring crosstalk between nociceptors and immune cells after discussions with her colleague and one of the new paper's senior authors, Aziz Moqrich. They both were interested in inflammation in the skin, from an immune and sensory neuron perspective, respectively.
“I had the intuition that the two systems are communicating with each other,” Ugolini told PRF. “At the time, very little was known about neuroimmune interactions.”
To determine if nociceptors affect immune responses during skin inflammation, the group used a mouse model in which nociceptors expressing the sodium channel Nav1.8 are ablated through expression of diphtheria toxin. To cause skin inflammation, these female mice were vaccinated with ovalbumin in complete Freund’s adjuvant (CFA); the investigators used CFA because nociceptor-deficient mice do not develop the hypersensitivity normally caused by this agent.
To their disappointment, the researchers found that the extent of inflammation and activation of different immune cells did not differ between control and nociceptor-deficient mice in this immunization-induced inflammation model. These results are included in the published study. However, “we could not exclude the possibility that the strong inflammation induced by CFA bypassed the neuroimmune regulation that could take place under more physiological conditions,” Ugolini said. The group thus continued exploring the role of nociceptors in other models of skin inflammation.
A viral skin inflammation model
Shortly after those investigations, a publication appeared on the role of nociceptors in the immune response to bacterial infection. These cells were found by Chiu and colleagues to play a role in regulating the innate immune response to Staphylococcus aureus infections (Chiu et al., 2013PRF news story). These findings encouraged Ugolini to look further into the role of nociceptors in skin inflammation.
She decided to test the role of these neurons in an infectious skin model in which it was possible to follow both the innate and adaptive immune responses. To that end, she spent a sabbatical in the laboratory of Francis Carbone, another senior author of the paper and an expert on HSV-1 infection.
“HSV-1 was the perfect virus for our research question. It produces skin lesions, infects sensory neurons, and induces a strong immune response,” Ugolini said.
HSV-1 is one of the most common cutaneous infections in humans, well known for the nasty and often itchy and painful blisters it causes. In the mouse model, HSV-1 goes through two phases of acute viral replication in the skin. In the first phase of infection, the virus infects keratinocytes, causing a skin lesion at the site of infection. The virus particles (virions) also infect sensory neurons, and spread by axonal transport to the sensory ganglia, where they undergo extensive replication. The virions then travel by anterograde transport along the many axons derived from the infected ganglia, until they reach the skin, where a secondary growth phase occurs, involving the entire dermatome innervated by the ganglia and resulting in an extensive lesion.
But from a methodological perspective, the problem with using nociceptor-deficient mice to study neuroimmune interactions during HSV-1 infection is that they might produce fewer virions, as they have fewer sensory neurons. Hence, any observed results might be confounded by differences in the amount of virus. Therefore, the researchers had to come up with a way to make sure the viral loads would be comparable in nociceptor-ablated and control mice. So the researchers came up with a solution.
“We engineered a virus deficient in thymidine kinase, an enzyme essential for viral replication in neurons but not in keratinocytes. Following infection with thymidine kinase-deficient forms of HSV-1, only the first phase of replication in the skin occurs. Additionally, the virus was engineered to also express an ovalbumin peptide, which we exploited to investigate the specific T cell response,” Ugolini explained.
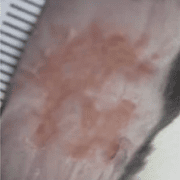
Administration of this HSV-1 strain to the flank produced skin lesions in both Nav1.8+ neuron-ablated and control mice. But while both groups had similar viral loads in the skin throughout the duration of infection, the lesions were larger and persisted longer in mice lacking nociceptors. This suggested that nociceptors inhibit the inflammatory response to HSV-1 skin infection, and thus promote tissue healing, which is impaired in the absence of nociceptors.
![Representative pictures of the skin lesion progression in control DTA [diphtheria toxin] (top line) and Nav1.8-DTA mice (bottom line). [p.i, post-infection]. Credit: Filtjens et al. Nat Commun. 2021 May 18;12(1):2936.](https://www.iasp-pain.org/wp-content/uploads/2023/02/Nociceptors.Viral_.Infections.InlineF.png)
Nociceptors regulate the innate immune response to HSV-1 infection …
To confirm that nociceptors indeed are capable of dampening the immune response to HSV-1 infection, the investigators determined what was happening in the skin lesions. They found that pro-inflammatory cytokines and chemokines accumulated in the lesioned skin of mice lacking nociceptors. To determine the cellular source of these inflammatory mediators, the group performed flow cytometry and intracellular staining on an array of skin cells. This revealed that both monocytes and neutrophils produced the pro-inflammatory cytokines tumor necrosis factor-alpha (TNF-α) and interleukin 1-beta (IL-1β) in nociceptor-deficient mice.
Next, the authors asked if nociceptors were involved in the recruitment of immune cells to the inflamed skin. While both nociceptor-deficient and control mice showed an increase in neutrophils, a type of white blood cell important in the innate immune system, in the first days of infection, high numbers of these cells persisted only in the skin of mice lacking nociceptors. This appeared to depend on prolonged recruitment of neutrophils to the skin, as cell death of neutrophils was comparable between nociceptor-ablated and control mice. Importantly, neutrophil depletion was sufficient to reduce skin lesion size to that observed in control mice.
In short, the above findings suggested that nociceptors control the innate immune response to HSV-1 infection, in particular by dampening the release of inflammatory cytokines by monocytes and by limiting neutrophil infiltration once HSV-1 virions are cleared from the tissue, and hence promoting skin regeneration.
… and the adaptive immune response, too
Next, Anais Roger, co-first author along with Jessica Filtjens, was interested in learning if the adaptive immune response might also be regulated by nociceptors. Indeed, the number of antigen-presenting Langerhans and dendritic cells (DC) – immune cell types important for the adaptive immune response – were reduced in the skin of nociceptor-deficient mice. Further, while the migration frequency of these cells to the draining lymph nodes, where they travel to, from the skin in order to present antigen to T cells, was comparable between the two groups of mice, DCs isolated from nociceptor-deficient mice were unable to induce proliferation of cytotoxic T cells, an important component of the adaptive immune response in vitro.
Going a step further, the authors asked if nociceptors impaired the DC-mediated T cell response in vivo as well. They performed adoptive transfer of fluorescently labeled T cells that are specifically activated by the antigen ovalbumin, and then infected the mice with the ovalbumin-expressing HSV-1 strain.
In vitro, these T cells proliferate after exposure to exogenous ovalbumin. In vivo, one week after the infection, the T cells had expanded in the spleen and draining lymph nodes, but their proliferation was robustly reduced in nociceptor-deficient compared to control mice, recapitulating the in vitro findings. On the other hand, the number of T cells in the skin did not differ between the two groups, suggesting that nociceptors are not involved in cytotoxic T cell recirculation from the lymph nodes to the skin.
“Overall, the experiments concerning the activation of cytotoxic T cell response strongly suggest that viral antigen uptake or processing is impaired in the absence of nociceptors,” Anais said. The effect, at least in part, seemed to be mediated by neutrophils, as their depletion in nociceptor-deficient mice restored levels of skin DCs and T cell responses to those observed in control mice.
The authors' discovery of nociceptor regulation of neutrophils is consistent with previous studies. For instance, previous work from Chiu's lab found that Nav1.8+ nociceptors suppress neutrophil influx during S. aureus infection (Chui et al., 2013). Later on, this group showed that a subset of skin-innervating nociceptors expressing the ion channel TRPV1 suppresses neutrophil influx during infection with S. pyogenes, another bacterial pathogen (Pinho-Ribeiro, 2018).
On the other hand, in a model of psoriasis-like skin inflammation caused by exposure to imiquimod, nociceptors actually stimulated the recruitment of neutrophils by driving the production of interleukin-23 (IL-23) in DCs (Riol-Blanco et al., 2014). And, in the present study, the researchers found that nociceptors did not regulate the immune response in a model of cutaneous vaccination.
“This really indicates that the role of nociceptors in the immune system is context dependent,” Ugolini said.
Limitations and remaining questions
The study has some limitations, according to Chiu.
“Killing off Nav1.8+ neurons during development through diphtheria toxin expression might dysregulate homeostasis, either of the nervous system or of the skin,” he said.
Nav1.8 is expressed in a large population of sensory neurons – about three-quarters, in fact. “It would be interesting to determine if a particular subset of nociceptors is responsible for the observed immune regulation during herpes simplex infection,” Chiu said.
Another unknown, according to Chiu, is that “it really is not clear by which mechanism nociceptors regulate the immune response to HSV-1 infection. Are they signaling to neutrophils and cytotoxic T cells directly, or are they communicating through other skin-resident cells? What molecules mediate this communication?”
Along these lines, Chiu's lab has previously shown that the neuropeptide calcitonin gene-related peptide (CGRP) released by peptidergic nociceptors during bacterial skin infections suppresses neutrophil recruitment. And in another recent publication by Ugolini and colleagues, the group identified TAFA4 as a neuropeptide that could potentially regulate the immune response to skin inflammation (Hoeffel et al., 2021). In that study, they found that following sunburn, nonpeptidergic C-fibers produce TAFA4 in the skin, promoting the production of IL-10 by dermal macrophages, which in turn leads to reduced skin inflammation and to tissue regeneration. Roger is currently investigating if TAFA4 could be the neuropeptide responsible for immune regulation in the HSV-1 infection model.
“Our ultimate aim is to identify and exploit the pathway that regulates the immune response, to better treat skin infections and inflammatory diseases,” Ugolini said.
PRF Correspondent Larissa de Clauser is a postdoctoral fellow at Eurac, Bolzano, Italy.