Pain researchers are well aware that microglia play a critical role in pain sensitization after nerve injury. Known as the immune cells of the central nervous system, microglia drive the neuroinflammatory signaling in the spinal cord that is characteristic of chemotherapy-induced peripheral neuropathy (CIPN) and other neuropathies. Now, a new study uncovers a novel mechanism underlying this neuropathic pain pathology and presents a promising therapeutic approach to alleviate the pain.
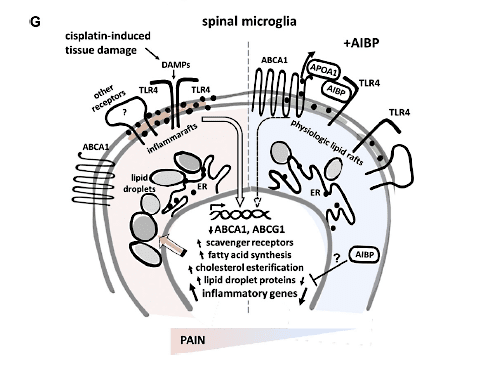
The new research from Yury Miller, Tony Yaksh, and colleagues at the University of California, San Diego, La Jolla, US, shows that cholesterol-rich lipid rafts in the plasma membrane of microglia drive spinal neuroinflammation in CIPN. These rafts are the sites where inflammatory receptor complexes assemble, including the formation of homodimers by Toll-like receptor 4 (TLR4). The investigators term these complexes “inflammarafts.” Depletion of cholesterol in spinal microglia turned the inflammarafts into more physiological lipid rafts, which reversed neuropathic pain in the CIPN model.
“The finding that cholesterol regulates microglial signaling is very exciting and novel, especially in the context of neuropathic pain,” said Ru-Rong Ji, Duke University School of Medicine, Durham, US, who was not part of the study. “This study also confirms that TLR4 is an important target to treat neuropathic pain.”
The study appeared July 5, 2021, in the Journal of Experimental Medicine.
Inflammarafts: A central motif of inflammatory signaling in microglia
The new research stemmed from an unlikely collaboration, according to Yaksh, co-senior author along with Miller.
“This collaboration is an example of: Think again about seminars you don’t want to go to," said Yaksh.“Yury Miller was giving a talk about cholesterol and atherosclerosis, and I wasn’t really interested at first. But then Yury got me excited about the role of cholesterol and lipids in inflammation, especially after showing how cholesterol could modulate the cellular response to inflammation. We then started collaborating, working with the ApoA-I binding protein (AIBP) he was studying in atherosclerosis that regulated membrane cholesterol and bound to TLR4, blocking its action.” Yaksh told PRF (see Woller et al., 2018).
In the new paper, the researchers investigated whether chronic pain is associated with altered cholesterol dynamics in spinal microglia, using CIPN as a model of neuropathic pain in mice. In this case, the animals received cisplatin, which caused severe tactile allodynia three days after injection of this chemotherapy drug.
Beginning to see a link between TLR4, cholesterol, and lipid rafts, the investigators found that mice with CIPN displayed increased formation of lipid rafts in spinal microglia. There was also increased dimerization of TLR4 in the plasma membranes of the cells.
“Lipid rafts are cholesterol-rich areas of the plasma membrane,” explained Miller. “They act like solid platforms where many proteins assemble and create functional complexes. An important protein on these rafts is TLR4, which needs to dimerize in order to trigger neuroinflammatory signaling. If there is too much cholesterol in the plasma membrane, which is common in inflammation, more lipid rafts form. Because of this link to inflammation, we suggested the term ‘inflammarafts.’ More inflammarafts mean more inflammatory signaling. In microglia, we thought this would lead to sensitization of neurons and sensitization to pain,” according to Miller.
Disrupting the inflammarafts reverses neuropathic pain
To see if that was true, the authors asked whether manipulating microglia cholesterol dynamics would prevent TLR4 dimerization and reduce neuropathic pain behaviors in mice. They injected AIBP intrathecally into the CIPN mice. AIBP works by removing cholesterol locally, near TLR4 receptors in the inflammarafts. The thinking was that less cholesterol would mean normalization of inflammarafts and no TLR4 dimerization.
Indeed, AIBP reversed tactile allodynia in CIPN animals and reduced TLR4 dimerization. This suggested that AIBP specifically targets inflammarafts, leading to reduced neuroinflammation and reduced neuropathic pain.
“AIBP was remarkable in reversing this allodynia, and the therapeutic effect of a single dose of AIBP lasted for months; we tested out to two months. This is unique, as no other agent can cause a complete reversal of neuropathic pain for such a long time,” Miller said.
AIBP reprograms microglia
While the therapeutic effect of a single dose of AIBP lasted for two months, the injected AIBP was only present in the lumbar spinal cord for a few hours.
“This means that AIBP is reprogramming the microglia, possibly by causing gene expression changes. Our next questions were: What happens with the gene transcription program? Does CIPN cause gene expression changes in microglia that AIBP then reverses?” said first author Juliana Navia-Pelaez.
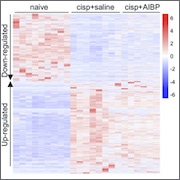
To answer those questions, the authors used RNA sequencing and differential gene expression analysis tools to compare gene expression changes in spinal microglia isolated from three groups of mice: naïve control mice, CIPN mice, and CIPN mice treated with AIBP.
CIPN caused widescale alterations in microglial gene expression. Among the genes that changed in response to CIPN were those involved in metabolism, inflammatory signaling, and homeostatic markers. CIPN also altered membrane cholesterol regulation, for example, by downregulating the cholesterol transporter genes Abca1 and Abcg1, indicating how nerve injury might cause specific gene expression changes that drive inflammaraft formation and microglial lipid dysregulation.
Changes in a group of genes upregulated in CIPN mice were reversed by AIBP. Of note, AIBP reversed inflammatory gene expression changes caused by CIPN, such as alterations in genes that encode cytokines like CCL2 and CXCL2. Similarly, AIBP reversed changes in gene expression caused by CIPN for genes involved in cholesterol metabolism, homeostasis, and lipid signaling that were related to the regulation of lipid droplets induced by CIPN in microglia.
Overall, these findings indicated that AIBP substantially rebalances neuroinflammatory pathways in spinal microglia after nerve injury by reprogramming altered gene expression that had been caused by damage to nerves.
Cholesterol: A new player in pain sensitization
So far, the authors had shown that TLR4 signaling in inflammarafts in spinal microglia drives CIPN. But how does baseline cholesterol signaling at the plasma membrane contribute to normal nociception? Does excess cholesterol, on its own, in the plasma membrane drive pain behavior in the absence of injury?
To find out, the authors focused on ABCA1 and ABCG1, two proteins that transport excess cholesterol out of the cell. They generated a microglia-specific ABCA1 and ABCG1 double-knockout (KO) mouse, which had the effect of increasing cholesterol in the plasma membrane of spinal microglia.
Remarkably, these microglia-specific double KO mice developed allodynia even without any stimulatory challenge or prior injury. This correlated with increased TLR4 dimerization and more inflammarafts in microglia.
“This is one of the striking findings in the paper. It shows that cholesterol buildup in spinal microglia alone is sufficient to drive neuroinflammation and pain behaviors,” Navia-Pelaez told PRF.
In subsequent experiments, the authors found that AIBP couldn't fully reverse CIPN-induced allodynia in ABCA1/ABCG1 double KO mice, unlike in their wild-type litter controls. This showed that AIBP partially requires cholesterol transporters to modify inflammaraft dynamics in microglia and reverse allodynia.
To pry a little deeper into this mechanism, the group looked at gene regulation changes induced by AIBP in the microglia of the double KO mice and in wild-type animals. In the absence of cholesterol trafficking machinery, AIBP failed to downregulate inflammatory genes, and also upregulated genes in cholesterol metabolism pathways. For example, the master cholesterol metabolism regulator gene Srebf2, which was downregulated in wild-type mice by AIBP, was upregulated in the KO mice. This indicates that AIBP induction of anti-inflammatory genes in microglia depends on cholesterol transporters.
TLR4 is the missing link
While the experiments showed that removing cholesterol from cholesterol-rich lipid rafts was important for the therapeutic effect of AIBP, the authors speculated that TLR4 might also be required. So they generated a microglial-specific TLR4 KO mouse. Not surprisingly, TLR4 KO mice had less severe allodynia in a CIPN model, suggesting that TLR4 expression in microglia plays a role in pain sensitization caused by cisplatin.
To further investigate the role of TLR4 in AIBP’s mechanism of action, the researchers created a mutated AIBP protein that could not bind to TLR4 (mutAIBP) and tested whether this mutant AIBP was still able to reverse allodynia in the CIPN mouse model.
“When we administered mutAIBP that lacks its TLR4 binding domain,” explained Navia-Pelaez, “it only modestly reduced neuropathic pain, as mechanical thresholds didn’t reach baseline levels, and the effect only lasted for a few days, as opposed to the months-long therapeutic effect of the AIBP that binds to TLR4.”
Overall, the research shows that AIBP can powerfully reverse CIPN by selectively depleting cholesterol from TLR4-containing inflammarafts and by reducing cytosolic lipid deposits in spinal microglia. The authors also hypothesize that inflammarafts might be a key motif that defines the inflammatory state of other cell types.
“Although this paper is focused on microglia, we think that inflammarafts or similar lipid raft domains might be a crucial motif for the activated state of a cell, be it microglia, neurons or other cell types. We are working on this idea. This changes the way we think about cholesterol in the membrane – it shows us that it is organizing all these proteins like TLR4 that change the state of the cell, and in this context, drive mechanical allodynia,” said Yaksh.
Indeed, since lipid rafts modulate ion channels such as NMDA receptors and sodium channels (Dart, 2010), the effects of inflammarafts might go well beyond the context of microglial inflammatory signaling in chronic pain.
Finally, Yaksh and Miller are keen to move the research forward into clinical translation. They are the scientific co-founders of Raft Pharmaceuticals, a startup company developing pain therapeutics targeting lipid rafts.
Fred Schwaller, PhD, is a freelance science writer based in Germany.
Credit: Featured image (top of page) from J Exp Med. 2021 Jul 5;218(7):e20202059. ©Navia-Pelaez et al.