Which cells are responsible for painful cold sensing in teeth – the sensation that can accompany eating ice cream, drinking iced tea, or having an infected tooth? And which signaling molecules do they use to carry out this function? Despite decades of research, full answers to these questions have remained elusive. A new study now proposes that a member of a familiar family of ion channels, on one of the main cell types in teeth, underlies the phenomenon of tooth cold pain.
Researchers led by Katharina Zimmermann, Erlangen University Hospital, Friedrich Alexander University of Erlangen-Nuremberg, Germany, report that odontoblasts, the tooth cells that make dentin, the tissue that sits below the tooth enamel, signal cold pain in teeth via the TRP channel TRPC5. With an impressive breadth of experimental techniques – ranging from electrophysiological recordings to knockout mice missing cold-sensitive ion channels, from pharmacological inhibition to an examination of human teeth – the investigators thus propose the odontoblast as a primary sensor of cold pain in teeth.
“A particular strength of this study is its multimodal approach,” said Barry Sessle, University of Toronto, Canada, referring to the study's combination of in vitro experiments, in vivo work in mice, and investigation of healthy and inflamed human teeth. And, while other TRP channels had been implicated previously in tooth cold sensitivity, “the focus on TRPC5 is novel and very exciting,” according to Sessle, who was not involved with the new work.
However, Sessle cautioned that there are fundamental questions that need to be addressed before the authors can clinch their case. For instance, among several other key uncertainties, he said it's possible that TRPC5 on dental afferent nerve fibers, rather than on odontoblasts, may be involved, as the authors themselves acknowledge. So the endings of those afferents could potentially be the actual primary cold sensors.
The work from Zimmermann and her fellow co-senior authors Jochen Lennerz, Massachusetts General Hospital and Harvard Medical School, Boston, US, and David Clapham, Boston Children's Hospital and Harvard Medical School, US, appeared March 26, 2021, in Science Advances. (See related PRF news story on another recent TRPC5 paper showing a role for the channel in mechanical and spontaneous pain.)
Previous theories
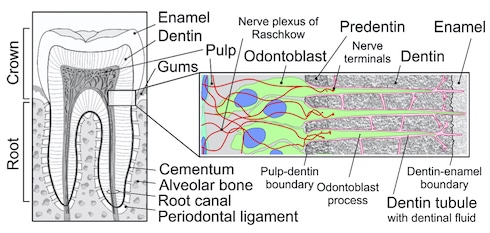
The study of cold pain in teeth goes back many decades, with a number of theories proposed to account for it. One theory has held that nerve fibers extending from the tooth pulp into tiny tubules within the dentin would respond to stimuli applied to the enamel, the hard tissue above the dentin that makes up the crown of the tooth.
A different theory has held that movement of fluid in the dentinal tubules, in response to a stimulus like a change in temperature, would activate nerve endings at the border between pulp and the tubules. In this scenario, fluid displacement would act as a mechanical stimulus that would somehow activate nerve fibers.
A third hypothesis has been that odontoblasts, which extend a process into the extracellular fluid-containing dentinal tubules, would detect mechanical distortion of the fluid and in some fashion pass a signal on to nearby nerve fibers. The discovery of the presence of TRP channels in odontoblasts would make that argument a defensible one.
“The idea was that since the odontoblast with its process is right in the dentinal fluid, it would have a large area of contact with the fluid and move in registry when the fluid moves in response to some thermomechanical distortion. That would probably make it more likely that the odontoblast is the cold pain sensor. And this idea came up with the proof of expression of TRP channels in the odontoblasts,” said Zimmermann.
Keeping their focus on odontoblasts, the researchers decided to take a more detailed look specifically at TRPC5, based on their previous work showing that this channel was sensitive to innocuous cold in heterologous cells; they had also found that the channel was present in the peripheral nervous system (Zimmermann et al., 2011). But they were unable to find any physiological function for the ion channel in noxious cold sensing.
“We worked to find out if there was any physiological role for TRPC5 in cold sensing in the skin, like in nociceptors in the skin, for example, or in thermal receptors, or in DRGs in culture. But really, we couldn’t find anything,” Zimmermann said. “My old friend from medical school, Jochen Lennerz, was in Boston at that time, and with David Clapham, who was my postdoc supervisor, we were thinking about where else TRPC5 could be a cold sensor.” This led them to teeth.
“We came to the conclusion that teeth are really incredibly cold sensitive. At that time, Jochen Lennerz was looking in teeth and using an antibody to stain TRPC5. He found that TRPC5 is in human teeth and greatly upregulated in inflamed human teeth. But at that time when we knew it was in teeth, it still didn’t mean anything physiologically for cold transduction. It just gave us a hypothesis,” said Zimmermann.
Testing the hypothesis
The researchers, including co-first authors Laura Bernal and Pamela Sotelo-Hitschfeld, began by looking at knockout mice missing TRPC5, TRPA1, or TRPM8. The last two TRP channel family members had previously been implicated in painful cold sensing in the skin, but their role in tooth cold pain had remained unclear.
Using a mouse dental pulp injury (DPI) model characterized by inflammatory pain, the researchers saw an increase in sucrose consumption in injured wild-type animals compared to uninjured animals (this increase is thought to reflect painful DPI in mice). But, while TRPA1 and TRPM8 knockout mice showed similar increases in sucrose intake after injury, the TRPC5 knockouts consumed sucrose to the same level as the uninjured controls (similar to the effect that the anti-inflammatory pain reliever indomethacin is known to have on sucrose consumption during dental injury pain in mice). From these results, the researchers concluded that TRPC5 plays a role during inflammatory tooth pain.
To learn more, the researchers next turned to an ex-vivo mouse jaw-nerve preparation. Here, they used suction electrodes to record electrical activity from the inferior alveolar nerve, which innervates the teeth, in response to cold stimuli applied to the jaw-nerve preparation.
The recordings revealed large responses to cold in about 10% of A- and C-fibers. The group noted that these cold responses were much larger than those seen in mouse skin nociceptors, based on comparisons of the findings to those from a skin-nerve preparation. Neurons from the jaw-nerve preparation also exhibited a lower threshold temperature of activation than the skin cold nociceptors.
The relative contributions of TRPC5, TRPM8, and TRPA1
The investigators next used TRP channel blockers to delineate the relative contributions of the three ion channels they had set their sights on. Two TRPC5 inhibitors together blocked about 60% of cold-sensitive tooth nociceptive responses in the jaw-nerve preparation. The addition of a TRPA1 blocker eliminated all tooth cold responses in fibers not blocked by one of the TRPC5 inhibitors.
“The interesting part was really when we looked with the pharmacological compounds because TRPC5 blockers only blocked around half of the cold responses,” said Zimmermann. “But when we added a TRPA1 blocker, it completely blocked everything. So it seemed like there could be a chain of events, like a network, where the electrical signals are going through and TRPA1 somehow sits at the end, and if you block TRPA1, all the cold responses are gone. But we don’t have an answer to this yet.”
Looking at the TRPC5 knockouts, the team saw about 50% reductions in the number of cold nociceptors, with the remaining nociceptors showing higher peak firing rates compared to all the other mouse strains along with high threshold temperature activation. But modulating TRPM8 pharmacologically had no effect on tooth cold responses. Further evidence for a role of TRPC5 and TRPA1 in tooth cold pain came from double knockout animals missing both ion channels. That experimental manipulation decreased the number of cold responses by one-third in the jaw-nerve preparation, and the remaining responses stayed much smaller.
The investigators next made in vitro recordings using cultured dental primary afferent neurons (DPANs), whose cell bodies are located in the cranial trigeminal ganglion. Interestingly, results from experiments using calcium imaging, and work using TRP channel modulators in various double knockout animals, indicated that DPANs sensitive to cold mainly used TRPM8 for cold responses, though TRPA1 and TRPC5 could also transduce cold in very few neurons.
On to the odontoblast
Further experiments showed that a very small percentage of DPANs from mouse trigeminal ganglia expressed TRPC5, and while the channel was functional, it only passed a small amount of current that was not affected by cold. So the next question was, What cell type within the tooth was the main contributor to the transduction of cold pain via TRPC5? The researchers turned their attention to the odontoblasts.
It turned out that nearly all of the odontoblasts at the border of the pulp and the dentin expressed the channel. The odontoblast processes within the dentinal tubules made contact with sensory axons at that border and were in tight association with the axons as the processes ascended into the dentinal tubules. Other cell types within the tooth, such as fibroblasts, were eliminated as contenders since they lacked TRPC5.
These results convinced the researchers that the odontoblast was the key cold sensor in teeth, but thus far all the experiments had been in mice. So the next task was to determine the relevance of TRPC5 in healthy and diseased human teeth.
The group first examined healthy adult human teeth that had been removed for orthodontic or cosmetic reasons, and found TRPC5 in the odontoblastic cell layer. TRPC5 was also present in sensory nerves at the border of pulp and dentin and in dentinal tubules, and at greater levels than TRPM8. Finally, TRPC5 expression in sensory nerves increased, and TRPM8 decreased, in human teeth that had inflammation of the pulp (pulpitis). This increased expression of TRPC5 extended to the degenerating dentin in the pulpitis model and the entire tooth root. The authors concluded from these findings that TRPC5 may be important for cold sensing in human teeth.
“The interesting part was that in mice we didn’t really see a lot of TRPC5 in nerves,” said Zimmermann. “But in human teeth, which are, of course, like a hundredfold larger than a mouse molar, there’s plenty of TRPC5 in the nerves. So in teeth, is it a combination of nerves and odontoblasts that sense cold? Or is it that, just during inflammation, TRPC5 expression increases in nerves because with inflammation, your tooth starts to become exposed and your nerves start to become exposed to the cold as well?” wondered Zimmermann.
Some major questions
Has the case for odontoblasts as primary cold sensors via TRPC5 been clinched? Not so fast, said Sessle, for a number of reasons.
First, on a technical note, he pointed to the anatomy of the mandibular incisor that the researchers used for some of their experiments, noting that the rodent mandibular incisor has been shown, in several studies decades ago, not to be an ideal model to study dental nociceptive processes since, unlike fully formed teeth, the pulp has a wide-open apex.
“So, for example, when you're recording extracellularly from afferents or trigeminal ganglion neurons supplying that open apex, you can't be sure exactly where those afferents are ending in relation to that tooth, that is, whether they're supplying dentin or the tooth pulp itself, or supplying some of the surrounding tissues,” Sessle said. Use of a different rodent tooth model that doesn't have this open apex would thus be a better model, Sessle said, to be more certain that the afferents being recorded from are in fact innervating the tooth pulp or dentin. To the investigators’ credit, Sessle also noted, some of the extracellular recordings were from afferents supplying molar teeth, which do not have this anatomical feature.
In terms of the bigger picture, Sessle also wondered whether cold sensing might not only depend on odontoblasts, since the authors have shown that TRPC5, and TRPA1 and TRPM8 are also expressed in the tooth afferents.
“Many of those nerve fibers are actually getting into the dentinal tubules. So when a cold stimulus is applied to enamel or to dentin, it could be hypothesized that those nerve fibers would become activated first, before the odontoblast,” Sessle said. “Although unlikely, given its expression in odontoblasts, it is possible that the TRPC5 in odontoblasts might have no functional role at all, or they might play a functional role more in the later stages of cold sensitivity.”
The definitive experiment for documenting if TRPC5 (or other TRP receptors) in odontoblasts are crucial in dental cold sensitivity, Sessle noted, would be to record in situ the activity from TRPC5-expressing odontoblasts themselves, and ideally also dental nerve endings, to see if they are cold sensitive and if the sensitivity could be blocked by TRPC5 inhibitors (or other TRP inhibitors). But it is technically challenging to record from these cells in situ because of their close proximity to dentin; this difficulty is something that has “plagued this field for decades,” he said.
Finally, Sessle noted that even if odontoblasts do prove to be the primary cold sensors, exactly how they would activate nerve fibers is unclear. The recent literature on this topic points to several mediators released from odontoblasts, as well as TRP and other receptors on the nerve endings, as players in nociceptive processes in the tooth.
“These are challenging experiments,” said Sessle. “I'm not trying to minimize their difficulty, but these are important issues and questions to address, if you really want to nail down exactly how, in this case, cold activates the afferents. I am hoping that this team of talented investigators will build upon their exciting new findings and use their extensive array of multidisciplinary skills to overcome the technical challenges and clarify exactly how the nerve endings are activated, and the role of odontoblasts, TRPC5, and other processes in the activation.”
Despite the open questions, the authors say their results show that researchers must expand their view of cold sensing in teeth.
“Probably our view on dental nociception was blinded or blurred so far because we always thought it’s the nerves" that matter, said Zimmermann. “The odontoblast could really be a primary sensory cell for nociception.”
Neil Andrews is a science journalist and executive editor of PRF.
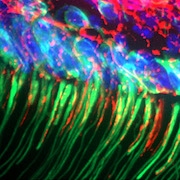
Featured image: TRPC5 (green) in tight association with sensory nerves (red, ßIII-tubulin), from TRPC5 reporter mouse molar tooth. From Bernal et al. Sci Adv. 2021 Mar 26;7(13):eabf5567.