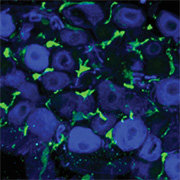
Immune cell recruitment and activation after nerve injury contribute to neuropathic pain, and recent studies have aimed to identify the types of immune cells and neuroimmune interactions that play a role in pain. Macrophages are one candidate cell type, but previous investigations showing their role in injury-induced hypersensitivity have been inconsistent.
Now, new research led by Allan Basbaum, University of California, San Francisco (UCSF), US, shows that macrophages in the dorsal root ganglia (DRG), but not at the peripheral nerve injury site, contribute to the initiation and persistence of mechanical hypersensitivity. Specifically, using a genetic mouse model allowing for macrophage depletion, Basbaum and colleagues show that depleting these cells prevents mechanical hypersensitivity immediately after nerve injury and reverses hypersensitivity 28 days after injury. However, these effects were not seen when macrophage depletion was localized to the nerve injury site.
Further, both male and female mice displayed anti-allodynic effects after macrophage depletion. However, the extent and mechanism of macrophage expansion in the DRG were sexually dimorphic.
“I think there are several really striking observations here,” said Andrew Shepherd, University of Texas MD Anderson Cancer Center, Houston, US, who was not involved with the study. “It adds key extensions to prior observations that macrophages are contributing to hypersensitivity in this kind of model, and it advances our understanding of where and what macrophages could be doing to foster this increase in sensitivity.”
The study was published January 14, 2020, in Nature Communications.
A contradictory literature
For several years, Basbaum’s lab has been investigating neuroimmune mechanisms in neuropathic pain. In 2016, they showed that after spared nerve injury (SNI), sensory neurons produce and transport the cytokine colony-stimulating factor 1 (CSF1) to the dorsal horn of the spinal cord, where it stimulates microglia proliferation (Guan et al., 2016PRF related news story). These findings provided evidence that sensory neurons influence microglia, the resident macrophages of the central nervous system (CNS), in the SNI model, but the researchers did not look at whether this CSF1 mechanism applied to peripheral macrophages.
The current study is “a natural follow-up to our work on the CSF1 story,” Basbaum told PRF. “CSF1 is induced in the sensory neurons, and there are macrophages in the DRG, but we can’t rule out the possibility that there’s a connection with the macrophages.”
To examine the role that macrophages play in neuropathic pain, Basbaum’s group used the macrophage Fas-induced apoptosis (MAFIA) genetic mouse model, in which macrophages can be depleted after administration of AP20187 (AP), a molecule that causes apoptosis. This model allows for depletion of macrophages specifically in the periphery, giving it an advantage over other methods of macrophage ablation.
Previous studies had produced conflicting results about whether macrophages are required for nerve injury-induced mechanical hypersensitivity. This is likely due to the variability and lack of specificity of methods to deplete macrophages, such as administration of the drug clodronate or viral ablation. A more recent study employed the MAFIA model and showed that elimination of peripheral macrophages reduced mechanical hypersensitivity from nerve injury (Shepherd et al., 2018PRF related news story). That study used a dose of AP that left DRG macrophages intact, leading to the conclusion that the macrophages in the periphery had a more crucial contribution to the hypersensitivity than those in the DRG.
First author of the current study, Xiaobing Yu, also at UCSF, commented, “We know after nerve injury there is significant macrophage expansion at the injury site and in the DRG, so the simple question is whether this is critical for mechanical allodynia. In the literature, people have started to tackle this topic, and unfortunately there are a lot of inconsistent findings.”
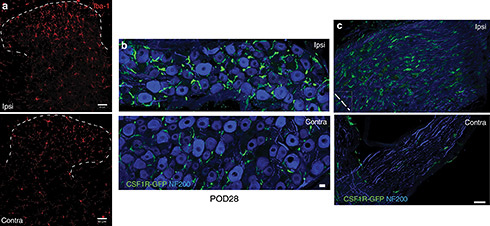
Considering the discrepancies, Yu, Basbaum, and colleagues set out to reexamine the question using the MAFIA mice. After confirming that SNI resulted in macrophage proliferation in the DRG ipsilateral to the injury site, the researchers tested the effects of depleting macrophages in the animals. They established a three-day protocol for administering AP and found that the dose they used significantly reduced the number of macrophages in the DRG without affecting the spinal cord microglia population.
Next, to test the effects in the context of nerve injury, the investigators administered AP for three days prior to SNI. This prevented the macrophage expansion in the ipsilateral DRG. Further, the macrophage-depleted mice did not develop hypersensitivity until at least seven days post-injury. Remarkably, depleting macrophages 28 days after SNI reinstated the anti-allodynic effect. Although this effect was transient, it indicated that macrophages also have an important role in the persistence of hypersensitivity.
Macrophages … but where?
The question remained of whether macrophages in the DRG or those at the injury site contributed to the hypersensitivity. The researchers addressed this question using a localized macrophage depletion method. They implanted a cannula over the nerve injury site and titrated a dose of AP that eliminated macrophages at that site but left DRG macrophages intact. These mice developed the typical hypersensitivity seen after nerve injury, suggesting that depleting peripheral macrophages was inconsequential and that DRG macrophages were the major contributor to hypersensitivity.
Consistent with that finding, the researchers also tested the effects of the localized depletion method 28 days after SNI and found that, in this case, too, it did not affect ongoing mechanical hypersensitivity.
These results contrast with previous studies showing that macrophage expansion at the injury site contributes to neuropathic pain, and it is still unclear which view is correct.
“There are obviously circulating monocytes that we couldn’t rule out completely, but the most likely effect is from the DRG macrophages to get the hypersensitivity,” said Basbaum.
“I agree that the DRG macrophage [proliferation] looks like it’s a major contributor,” said Shepherd, “but there is still a role for the peripheral macrophage infiltration. Rather than truly excluding a contribution of peripheral infiltration to pain, it’s adding further weight to the claims that the DRG [proliferation] is a key contributor.” He added that replicating these experiments in other labs will help pain researchers come to a consensus.
What drives macrophage expansion in the DRG?
As mentioned above, Basbaum’s group previously found that injured sensory neurons produce CSF1 that is transported to the spinal cord dorsal horn, where it stimulates microglia proliferation. Macrophages in the DRG express CSF1 receptors, so the team investigated whether CSF1 was responsible for macrophage expansion after nerve injury.
Using mice in which CSF1 was deleted from sensory neurons, the researchers found that the lack of CSF1 prevented injury-induced macrophage expansion in the DRG in the male mice used for these experiments. This suggested that sensory neuron-derived CSF1 influences the proliferation of DRG macrophages.
The researchers also wanted to test whether the proliferating DRG macrophages influenced sensory neurons. They looked at expression of brain-derived neurotrophic factor (BDNF) in sensory neurons, as it has been shown to have pronociceptive effects. They found that SNI produced a fourfold increase in BDNF messenger RNA (mRNA) expression in DRG neurons, and that depleting macrophages in the MAFIA mice totally blocked that increase.
The loss of macrophages also prevented upregulation of the proinflammatory cytokine interleukin-1β (Il-1β) that is usually seen after nerve injury. Interestingly, though the researchers expected to see mRNA expression of Il-1β in the DRG neurons or in satellite cells, they only detected it in the DRG macrophages. They concluded that the anti-allodynic effect of depleting macrophages was due in part to a reduction in Il-1β.
The interactions among different cell types in the DRG are especially intriguing to Basbaum, and he suspects that these circuits may influence what happens in the dorsal horn.
After nerve injury, “things are happening quickly,” he said. “The CSF1 is induced easily within 24 hours, microglia start to proliferate within two days in the cord, and the macrophage changes can be seen pretty quickly. So it’s possible that they’re occurring in temporal succession, or they may even be completely independent,” according to Basbaum.
Don’t forget sex differences!
Recently, researchers have uncovered sexual dimorphism in the contribution of immune cells to pain (see PRF related news story). So Yu and colleagues also tested female mice to see if the results they saw in male mice held true. They found a significant increase in macrophages in the DRG after nerve injury, but this increase was not as robust compared to the male animals.
Interestingly, the expansion of macrophages in the DRG of female mice did not depend on CSF1, as it did in males, with the females displaying the same macrophage proliferation after deletion of CSF1 from sensory neurons.
Further, as in males, ablation of DRG macrophages in females prevented mechanical hypersensitivity after SNI and reversed hypersensitivity 30 days after SNI. This showed that the macrophage contribution to the initiation and persistence of post-injury hypersensitivity was not sexually dimorphic.
Shepherd, noting the finding that female mice had less macrophage proliferation in the DRG compared to males yet a similar degree of hypersensitivity, said that “more work needs to be done to look at the relative dependence of the pain sensitivity on the degree of macrophage infiltration.”
In terms of future research, understanding the different effects of targeting macrophages in the DRG versus those in the periphery, as well as testing the macrophage contribution to pain using different pain models, is necessary. Although SNI is a robust model of neuropathic pain, similar macrophage depletion experiments in more clinically relevant models will be helpful.
“The majority of neuropathy cases that are going to come into the clinic are probably not due to surgical trauma or battlefield trauma. It’s going to be overwhelmingly things like chemotherapy and diabetes, where you have a bunch of other comorbidities which can influence the immune system,” according to Shepherd.
Finally, Yu said that the use of human induced pluripotent stem cells (iPSCs) will provide the best path forward. Turning iPSCs cultured in a dish into sensory neurons “would be a great model to study pain biology and look at interactions between immune cells and human sensory neurons. The human iPSCs will potentially provide us a platform to do something we were not able to perform in the past.”
Sarah Najjar is a PhD candidate at the University of Pittsburgh, US.