Genetic mutations that produce congenital insensitivity to pain (CIP) have sparked enormous interest in the pain field in the last decade as they offer enticing clues about the pathophysiology of pain and point the way to new potential analgesic targets. Alterations in genes that code for ion channels, transcription factors, and receptors have emerged as critical contributors to CIP.
Now, a new case report shows that a loss-of-function mutation in the enzyme fatty acid amide hydrolase (FAAH), as well as a microdeletion in a new “pseudogene” downstream of the FAAH gene called FAAH-OUT, explained a near complete insensitivity to pain in a female patient. This individual had higher levels of the endocannabinoid anandamide and related molecules that are normally broken down by FAAH, highlighting the role of the endocannabinoid system in this instance of pain insensitivity.
“This is an absolutely fascinating case report of a patient who doesn’t feel pain, similar to the Nav1.7 story a few years ago, but this time in the endocannabinoid realm. It confirms a significant body of evidence from the preclinical literature that inhibition or genetic deletion of FAAH is antinociceptive,” said David Finn, National University of Ireland, Galway. Finn studies the role of the endocannabinoid system in pain, anxiety, and stress but was not involved in the work.
The case report, led by Devjit Srivastava, Raigmore Hospital, Inverness, UK, and James Cox, University College London, UK, was published online March 27, 2019, in the British Journal of Anaesthesia.
Painless surgeries
The new report describes a 66-year-old woman from Scotland who initially came to Srivastava’s attention after being referred to him for a trapeziectomy, a notoriously painful hand surgery, in 2013.
“The first thing she said to me was that she doesn’t feel pain, doesn’t need an anesthetic, and doesn’t take painkillers,” Srivastava told PRF. “As you can imagine, I was a little taken aback by this, but as an anesthetist you’re supposed to make sure that the patient feels no pain during and after surgery. I took no risk and gave her the normal anesthetic protocol, but what she had said stayed with me.”
Following up with the patient the next day, Srivastava discovered that she only received 1 gram of paracetamol (acetaminophen), which she had to be convinced to take; she had not taken any other analgesics, and reported no pain at all. This led him to suspect that he had a unique case on his hands.
Srivastava then reached out to Cox, a pain geneticist, to try to understand what, if any, genetic alterations might underlie the insensitivity to pain in this patient. Cox had previously characterized a mutation in SCN9A, a gene that encodes the voltage-gated sodium channel Nav1.7, in a Pakistani family with CIP (Cox et al., 2006).
Incidentally, Srivastava saw the patient again in 2018, since she was due for a similar procedure on her other hand. This gave him an opportunity to further investigate her assertion that she was pain free.
“Part of me was initially skeptical. I thought she may have just been on the high side of the pain threshold spectrum and we may be getting carried away with this. But when she came back for the second surgery, I didn’t give her any analgesics before, during, or after, only propofol and an inhalational anesthetic. I followed up the next day and discovered that she hadn’t taken a single painkiller, reported a pain score of 0/10 throughout, and was discharged with no problems. This episode finally convinced me of her phenotype,” said Srivastava.
First author of the current study Abdella Habib, whose background was in hormonal regulation of appetite, knew of Cox and the work he was doing in pain genetics from their time together at Cambridge University, UK. When Cox started his own lab at University College London, Habib joined him as a postdoc in 2012.
“I didn’t know anything about human genetics at the time but was well aware of the work James had done identifying families with inherited insensitivity to pain, so I thought it was a great opportunity, and I followed him to London,” Habib told PRF. Habib now leads his own research group at Qatar University, Doha.
The partnership has proven very fruitful, as previously Habib and Cox, along with colleagues, identified a novel pain-insensitive phenotype caused by a mutation in the transcription factor ZFHX2 in an Italian family. The phenotype was so unusual and unrelated to any gene previously associated with CIP that the researchers termed the condition Marsili syndrome, from the name of the family (Habib et al., 2018).
A novel combination
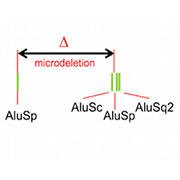
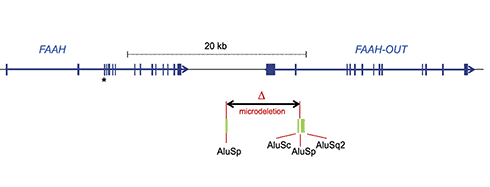
After extensive genetic sequencing and analysis, Habib, Cox, and colleagues identified two mutations underlying the pain insensitivity in the female patient. One was a hypomorphic mutation (which causes a partial loss of function), namely a single nucleotide polymorphism (SNP) in the gene that encodes the FAAH enzyme. The other was a rare, approximately 8-kilobase microdeletion downstream of the 3’ end of FAAH in a novel pseudogene (a DNA sequence that resembles a gene but has been mutated into an inactive form) that the researchers termed FAAH-OUT.
“The FAAH SNP is quite frequent in the general population, estimated to be at around 25 percent,” said Cox. “The FAAH-OUT microdeletion is much rarer. The patient’s son has it, as does as an anonymous male from Colombia, but neither have the FAAH SNP. This patient is the first person we are aware of to carry both mutations,” explained Cox.
The son has some diminished pain sensitivity but not to the same extent as his mother, while the phenotype of the Colombian male is unknown. FAAH SNPs are associated with a reduced need for postoperative analgesia in some cases (Cajanus et al., 2016), but as Habib explained, “it’s clear that both genetic alterations are required for this particular phenotype, making it a rare and previously unknown condition.”
Looking to the endocannabinoid system for an explanation
FAAH is an enzyme responsible for the breakdown of anandamide, an endocannabinoid known to elevate mood and reduce pain. Both FAAH and anandamide are part of the endocannabinoid system, which plays a role in a host of physiological responses, including pain. So the researchers turned their attention to that system to understand how the newly identified mutations might have contributed to the pain insensitivity phenotype of their female patient.
They measured blood levels of anandamide, 2-Arachidonoyglycerol (2-AG, a related endocannabinoid), as well as other substrates for FAAH including palmitoylethanolamide (PEA) and oleoylethanolamine (OEA). Compared to controls, circulating concentrations of anandamide were increased by 70 percent, and PEA and OEA levels were almost tripled, all of which suggests a loss of function of FAAH in the patient. Concentrations of 2-AG were unchanged, which is consistent with reports that 2-AG is mainly degraded by the enzyme monoacylglycerol lipase and not FAAH.
Another interesting aspect of the phenotype is the non-pain characteristics of the patient, including a cheerful disposition and frequent memory lapses, as well as low anxiety and fear levels. This is consistent with the preclinical literature, according to Finn.
“There is quite a strong body of evidence to indicate that inhibiting or deleting FAAH is anxiolytic [inhibits anxiety] in standard tests of anxiety in rodents,” Finn explained.
As for the memory deficits that the patient reported, this could relate to enhanced fear extinction or extinction of memories in general, both of which involve the endocannabinoid system.
“The endocannabinoid system plays a role in memory formation or facilitating extinction of memories. We see this in animal behavioral paradigms of memory where cannabinoids often impair memory, and we see this in humans where smoking cannabis can cause short-term memory deficits. The endocannabinoid system also plays an important role in regulating synaptic transmission and plasticity that relates to memory in brain regions such as the hippocampus,” Finn told PRF.
One for now, more to follow?
Although the patient in the new study is the first reported case of someone carrying both the FAAH SNP and the FAAH-OUT microdeletion, Cox believes it’s likely that there are others yet to be discovered who also carry both mutations.
“This FAAH-OUT microdeletion is flanked by repeat sequences, which are very similar Alu sequences [a DNA sequence that can move from one place to another within a genome; this is known as a transposable element or “jumping” gene], and it’s well known that in other genomic disorders these repeats can predispose to deletions or duplications. So I wouldn’t really be surprised if other individuals have a similar deletion as a consequence of these repeat sequences at each end of the deletion,” according to Cox.
Another reason to think there are similar individuals still to be identified is that the patient only came to the attention of medical professionals when she was already in her 60s.
“There are people out there who are pain free and who are not going to the doctor, and pain is such a common reason for initially visiting a doctor, so it’s difficult to find these people,” said Cox.
The new work has a number of additional implications. Perhaps most notably, it could build interest in the development of FAAH inhibitors as analgesics, as this case report essentially represents a human validation of a preclinical pain target.
In addition, targeting FAAH-OUT “could provide a new route for delivering pain relief by focusing on the gene that appears to regulate FAAH expression, possibly via a gene therapy route, instead of using small molecule inhibitors. That may be a better route to pursue,” said Habib. (Cox and colleagues recently used a technique called CRISPR interference to show that FAAH-OUT could regulate FAAH expression; this work, which has not yet been peer reviewed, was recently reported in bioRxiv; Mikaeili et al., 2019).
“There is potential for widespread clinical applicability in targeting FAAH and FAAH-OUT not just in terms of pain, but also for anxiety, depression, and their comorbidity, which is very high with chronic pain,” said Finn.
Finally, new reports like the current one spark great interest in the scientific and broader non-scientific communities, which could help overcome one of the challenges of studying CIP patients: They are exceedingly rare.
“In the last decade we have been working with only a handful of CIP patients,” said Cox, “but every time a new report like this comes out it helps us to recruit more. We now have upwards of 80 individuals who have emerged after this paper was released who have very similar phenotypes, and we are currently investigating them. We are very thankful for this particular patient. She has been really kind to work with us over these past few years and has shown great patience with us; we are really grateful.”
Dara Bree is a postdoctoral fellow at Beth Israel Deaconess Medical Center and Harvard Medical School, Boston, US.
Image credit: Habib et al., 2019