The experience of pain includes both sensory and affective components. Pain researchers have learned a lot about the former—about the peripheral mechanisms by which noxious stimuli are transduced into electrical signals that travel from the body to the dorsal root ganglia and then into the spinal cord. However, much less is known about brain circuits that transform this nociceptive information into an affective pain percept.
New research led by Grégory Scherrer and Mark Schnitzer, Stanford University, US, now uses calcium imaging, chemogenetics, and other techniques to identify a “neural ensemble” in the basolateral amygdala (BLA). This ensemble is active upon acute noxious stimulation to specifically encode the negative affective valence—the unpleasantness—of pain, with no impact on pain’s sensory component. The researchers further show that the BLA ensemble encodes pain unpleasantness in a model of chronic neuropathic pain.
“The power of this study lies with the utilization of numerous novel techniques to measure activity of circuit-level neurons in the context of nociceptive stimuli, in the amygdala of mice. Something like that had not been done before in the central nervous system in the setting of pain,” said Benedict Kolber, Duquesne University, Pittsburgh, US, who was not involved with the research.
“It is very interesting to see that certain neuronal ensembles are specifically activated by nociceptive stimuli, ultimately giving an organism the full flavor of pain in the context of affect,” Kolber continued. “From a therapeutic perspective, these findings could be extremely powerful, because if we could just get rid of the negative valence of pain, then even a patient with a 10 on an intensity pain scale could say it doesn’t bother them—that it’s a 1 on an unpleasantness scale—which would greatly increase their quality of life.”
The work was published online January 18, 2019, in Science.
A miniature microscope reveals a neural ensemble
“When I started my lab at Stanford University,” Scherrer told PRF, “I wanted to look at the affective aspect of pain and bring the best neuroscience techniques to the pain field. I was very fortunate because my office was right next to the office of Mark Schnitzer, a neuroscientist with expertise in imaging techniques who developed miniature microscopes for in vivo calcium imaging. This led to many conversations and ultimately a great collaboration investigating the affective components of pain in the amygdala.”
At this time, co-first author Greg Corder began a postdoc in Scherrer’s lab. For the new study, Corder, who is now a faculty member at the University of Pennsylvania, Philadelphia, US, co-first author Biafra Ahanonu, a former graduate student in the Schnitzer lab, and colleagues mounted one of Schnitzer’s miniature microscopes to the heads of freely behaving mice that had been engineered to express a calcium indicator in principal neurons of the BLA. This allowed the investigators to track somatic calcium levels, an indicator of neuronal activity, when the animals were exposed to diverse innocuous and noxious stimuli.
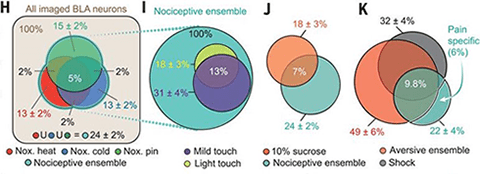
Depending upon the stimulus, the group observed calcium responses in approximately 13% to 15% of neurons in response to nociceptive stimuli applied to the hind paw, including noxious heat, cold, and pin prick. About 7% of neurons responded to innocuous light touch stimuli, a subset that did not overlap with the nociceptive neurons.
Further analysis revealed what the researchers termed the BLA nociceptive ensemble: an overlapping population of neurons in the BLA that encoded nociceptive information across different modalities, including noxious heat, cold, and pin prick. The ensemble consisted of 24% of active BLA neurons.
To test if the BLA nociceptive ensemble broadly encoded stimulus valence, the researchers presented the animals with sucrose, an appetitive stimulus. The investigators found that it did not, as neurons that encoded sucrose consumption only overlapped with a subset of the nociceptive ensemble.
Furthermore, approximately 6% of imaged BLA ensemble neurons were specific to naturalistic nociceptive stimuli and to no other aversive sensory stimuli including electric shock, repulsive odor, loud noise, bitter taste, and facial air puff. “We would have predicted that aversive stimuli in general and painful stimuli activate a similar subset of cells because both are associated with negative valence. Thus, this finding was very intriguing and surprising to us, and this is something that we want to look into further,” Scherrer said.
A causal link revealed by a chemogenetic approach
To see if the BLA nociceptive ensemble played a causal role in producing pain behavior, the researchers expressed an inhibitory designer receptor exclusively activated by designer drug (DREADD) in the ensemble, in genetically engineered, targeted recombination in active populations (TRAP) mice. Put simply, what this chemogenetic strategy allowed the group to do is silence BLA nociceptive ensemble neurons activated by a repeated pin prick stimulus. The team reasoned that silencing these cells would change how the animals behaved when exposed to all kinds of noxious stimuli, considering that the BLA encodes many types of nociceptive stimuli within a core ensemble.
They found that silencing the pin prick-responsive neurons with the DREADD agonist clozapine-N-oxide (CNO), which was injected subcutaneously, reduced both attending and escape behaviors of the mice, compared to vehicle-treated animals. But, the ability to detect the stimulus, as well as withdrawal responses to mechanical and heat stimuli, were left intact.
“Initially, we thought the experiments did not work because I did not observe any differences in the reflexive behaviors,” Corder said. “But since I recorded the behavioral responses of every animal with three cameras placed at different angles, we were able to see that the CNO-injected animals were not reacting ‘normally.’ We then repeated the experiment on the same animals and saw the same effect: The CNO-treated animals stayed calm and did not exhibit the same escaping behaviors or attending behaviors to the stimulated area, as compared to their vehicle controls.”
Corder also tested operant pain behavior using a thermal gradient assay. Here, vehicle-injected TRAP mice learned to avoid noxious heat and cold zones. But the CNO-injected animals visited the noxious zones more frequently and for longer periods of time. This suggested that these animals found the noxious zones less aversive. Additional experiments would show that silencing the BLA nociceptive ensemble had no effect on reward or anxiety responses. Together, the results suggested that, in response to acute noxious stimuli, the ensemble encodes the negative valence specifically of nociceptive information.
What about chronic pain?
Scherrer and colleagues hypothesized that allodynia and pain hypersensitivity characteristic of chronic neuropathic pain might stem from maladaptive transformations in BLA coding. So they tracked BLA neural activity in mice before and after sciatic nerve injury, a model of neuropathic pain.
The team found that a subset of BLA neurons stably encoded noxious mechanical and cold stimuli throughout the development of chronic pain. Nerve injury did not increase the number of spontaneously active BLA nociceptive ensemble neurons or of the overall BLA population.
Interestingly, the authors discovered that BLA neural activity in response to light touch now activated a greater number of cells in the BLA nociceptive ensemble in injured mice. Along with this came increased reflexive paw withdrawal responses and enhanced affective-motivational pain behaviors. These experiments showed that the BLA might play a role in the development of chronic pain.
Lastly, the authors tested if, by manipulating the BLA ensemble, they could prevent light touch from becoming aversive and eliminate the unpleasantness of chronic pain. To do so, they used TRAP mice in which they could specifically inhibit BLA ensemble neurons that respond to light touch using CNO. They implemented this “light touch TRAP” protocol 21 days after nerve injury, a time when allodynia had fully developed.
Vehicle-treated mice displayed allodynia and hyperalgesia for both reflexive and affective-motivational pain responses, compared to uninjured animals, when tested at 42 days post-injury. However, CNO treatment in injured animals had no effect on reflexive pain responses but did decrease affective-motivational behaviors, regardless of the stimulus intensity or modality.
The authors next looked at cold allodynia, which is also a problem in patients with neuropathic pain. Here they used a two-chamber thermal escape-avoidance assay where one chamber was cold while the other was kept at a neutral temperature.
As expected, the authors saw that uninjured mice that received the TRAP protocol stayed away from the cold chamber, as did, to a greater extent, injured mice who received saline. Remarkably, however, injured, CNO-injected mice seemed almost totally indifferent to temperature, spending equal time in the cold and neutral chambers.
All told, once again the results showed that the BLA nociceptive ensemble was necessary for the aversiveness of pain, this time in the context of chronic neuropathic allodynia and hyperalgesia.
“Overall I think this is a great paper that uses amazing techniques that will be very valuable to the pain field in the future,” Kolber said.
Open questions
One question for future research is whether the changes in the BLA nociceptive ensemble after injury are a result of peripheral or central sensitization, or could even be due to changes in input the amygdala receives from other brain regions or to changes in the amygdala itself.
Kolber also wonders about the role of stress. “How much does stress affect the outcomes? During the miniature microscope imaging, the authors used blocks of stimuli where the time between stimuli is listed as 60 seconds. Coming from the stress field, where people often wait a week before performing the next stress behavior experiment, 60 seconds does not seem like a lot of time to me. The time between stimuli does not necessarily confound the findings, but it is an important factor to consider.”
As for Corder and his lab, he said that “we want to identify other brain circuits that the amygdala talks to in relationship to pain affect. For example, we are looking at the BLA connection to the nucleus accumbens and how emotional information contributes to chronic pain and comorbid depression.”
Meanwhile, Scherrer’s lab, in collaboration with Schnitzer’s group, is now focused on recording neural activity from multiple brain regions simultaneously, for example, from the cingulate cortex and the amygdala, to determine how different brain circuits may work together to encode pain unpleasantness.
Also, “We want to figure out how the neurons in the BLA nociceptive ensemble function and look for specific molecular targets, such as membrane receptors or ion channels, expressed by those cells that can ultimately be targeted with drugs,” Scherrer said.
Francie Moehring, PhD, is a postdoctoral research fellow at the Medical College of Wisconsin, Milwaukee, US.
Featured image credit: Corder et al. Science. 2019 Jan 18; 363(6424):276-281.